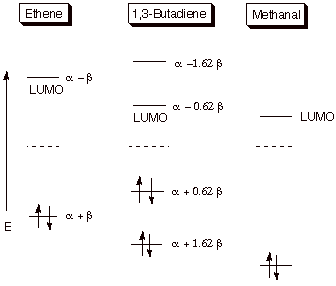Molecules | Free Full-Text | Synthesis, Physical Properties, and Reactivity of Stable, π-Conjugated, Carbon-Centered Radicals
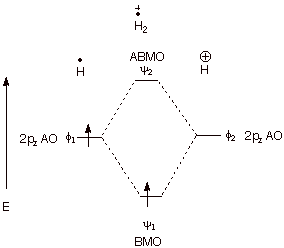
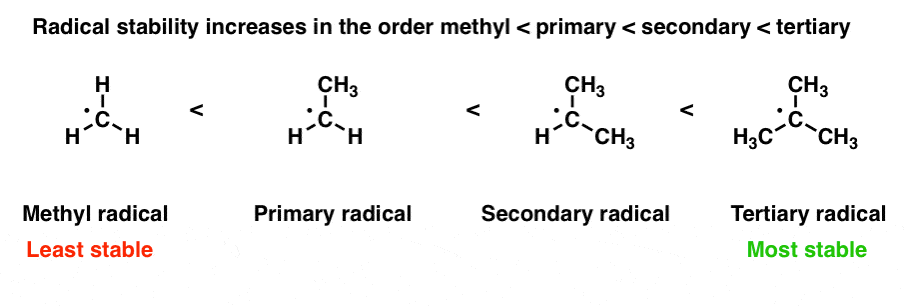
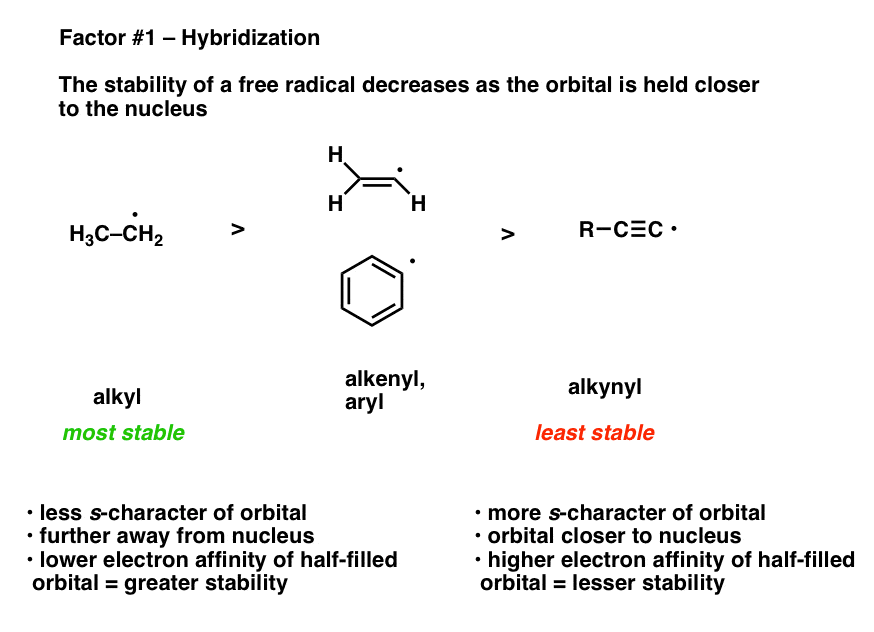
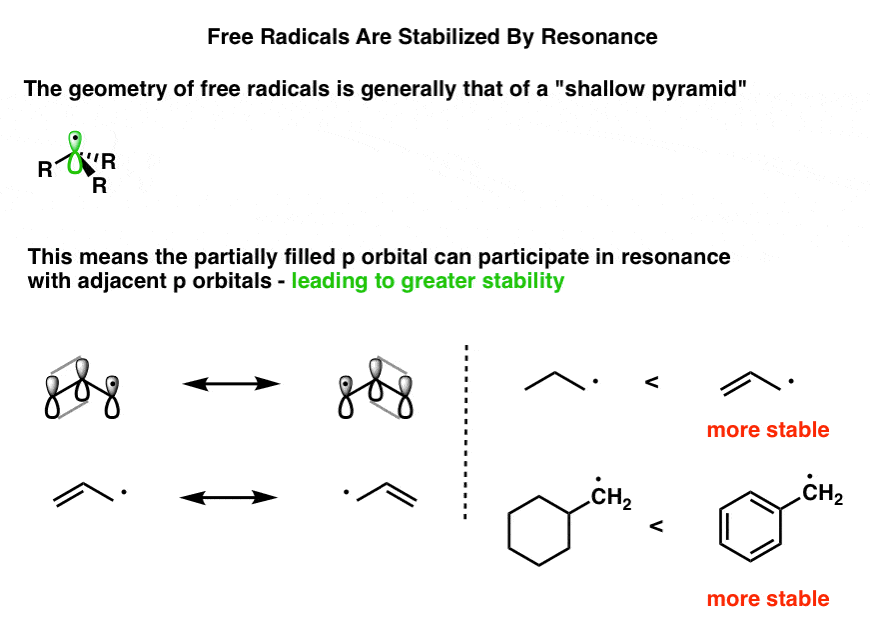
1. Strong Covalent Bonds. Consider the pi bond of ethene in simple molecular orbital terms (The qualitative results would be the same for any pi or sigma bond.

A Simple Approach to Achieve Organic Radicals with Unusual Solid-State Emission and Persistent Stability | CCS Chemistry

Depictions of the HOMO (red/blue) and LUMO (green/ yellow) of P, 1 + ,... | Download Scientific Diagram
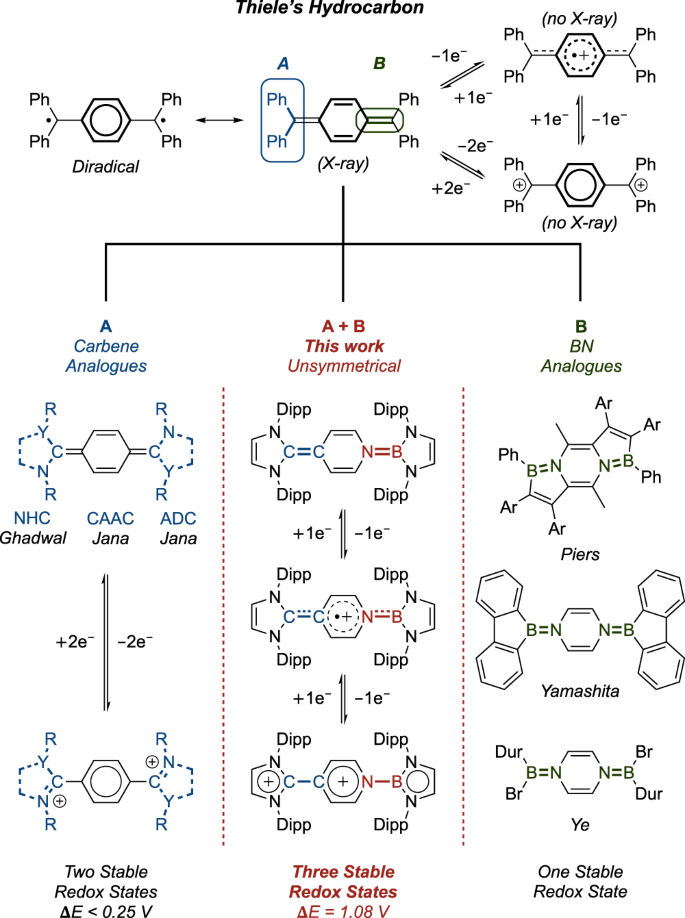
A crystalline radical cation derived from Thiele's hydrocarbon with redox range beyond 1 V | Nature Communications

Fluorescent Organic π‐Radicals Stabilized with Boron: Featuring a SOMO–LUMO Electronic Transition - Ito - 2022 - Angewandte Chemie International Edition - Wiley Online Library

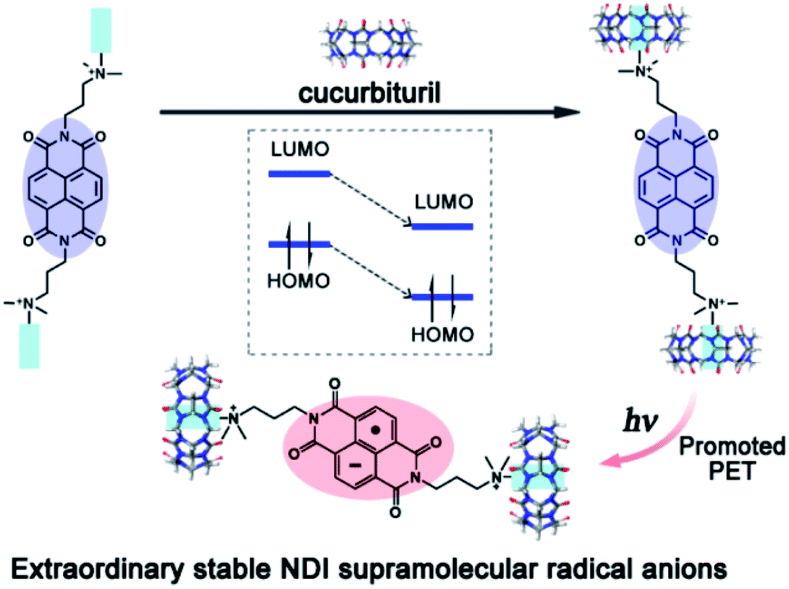
A supramolecular strategy for tuning the energy level of naphthalenediimide: Promoted formation of radical anions with extraordinary stability - Chemical Science (RSC Publishing) DOI:10.1039/C5SC00862J

Fluorescent Organic π‐Radicals Stabilized with Boron: Featuring a SOMO–LUMO Electronic Transition - Ito - 2022 - Angewandte Chemie International Edition - Wiley Online Library

Boron-centered stable radical anion/radical cation pair. Reproduced... | Download Scientific Diagram

Air-/Heat-Stable Crystalline Carbon-Centered Radicals Derived from an Annelated N-Heterocyclic Carbene | Journal of the American Chemical Society
Tuning the stability of organic radicals: from covalent approaches to non-covalent approaches - Chemical Science (RSC Publishing) DOI:10.1039/C9SC06143F

Highly Stable 1,2-Dicarbonyl Radical Cations Derived from N-Heterocyclic Carbenes | Journal of the American Chemical Society
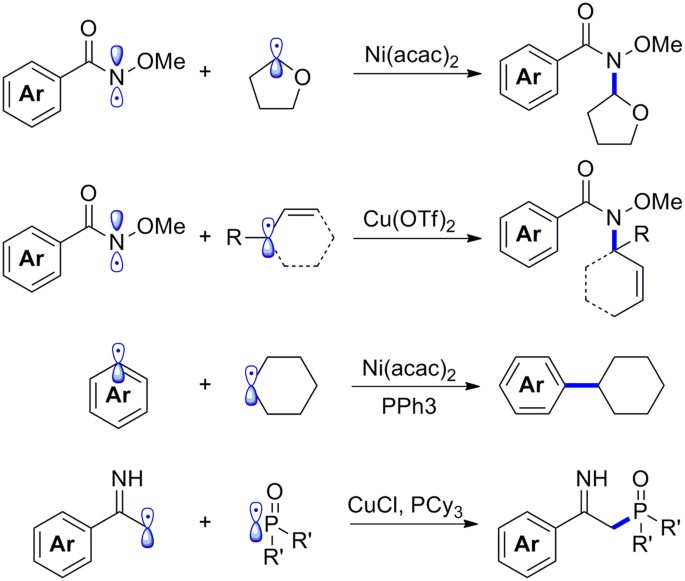
Stabilization of Two Radicals with One Metal: A Stepwise Coupling Model for Copper-Catalyzed Radical–Radical Cross-Coupling | Scientific Reports

Theoretical basis for the stabilization of charges by radicals on electrified polymers - Chemical Science (RSC Publishing) DOI:10.1039/C6SC02672A

SOLVED: CHAPTER Five Organic Chemistry EMiStRY Orbitale Combinations Table 5.3 Possible Frontier Molecular ( Examples of' Typical Qutcome HOMO LUMO ofS ! substitution Bond = formation in step or free radical addition