science chemistry redox reaction zinc hydrochloric acid | Fundamental Photographs - The Art of Science
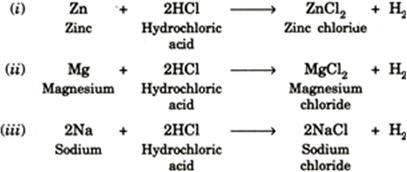
Write the balanced chemical equations for the reactions that take place when zinc, magnesium and sodium react with dilute hydrochloric acid. from Science Acids, Bases and Salts Class 10 UP Board

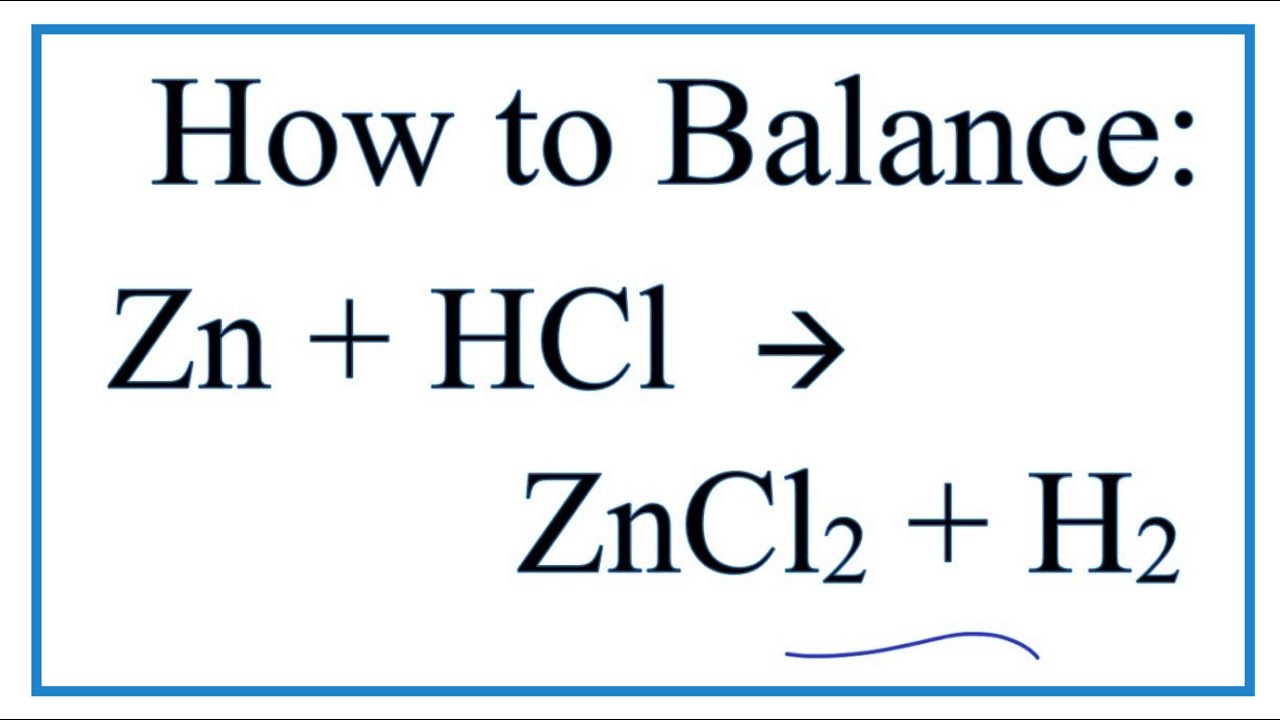
Zinc and hydrochloric acid react according to the reaction: Zn((s))+2HCl((aq.))rarr ZnCl(2(aq.))+H(2(g)) If 0.30 mole of Zn are added to hydrochloric acid containing 0.52 mole HCl, how many moles of H(2) are produced?

science chemistry redox reaction zinc hydrochloric acid | Fundamental Photographs - The Art of Science

SOLVED: Zinc metal reacts with hydrochloric acid according to the following balanced equation: Zn(s) + 2 HCl(aq) ? ZnCl2(aq) + H2(g) ?rH = -154 kJ mol-1 When a quantity of Zn is

SOLVED: Consider the reaction between zinc metal and hydrochloric acid. Which of the following would increase the rate of the reaction? 3 ZnClz 2 HCI Zn Hz using zinc powder instead of