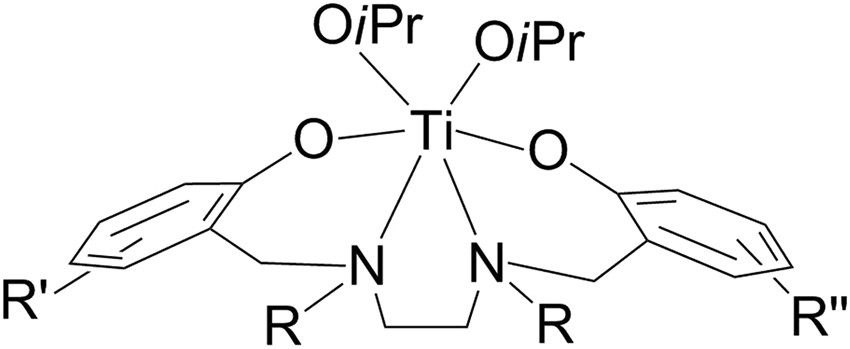
Synthesis of Pure Enantiomers of Titanium(IV) Complexes with Chiral Diaminobis(phenolato) Ligands and Their Biological Reactivity | Scientific Reports

Low cost Ti–Si intermetallic compound membrane with nano-pores synthesized by in-situ reactive sintering process | Scientific Reports
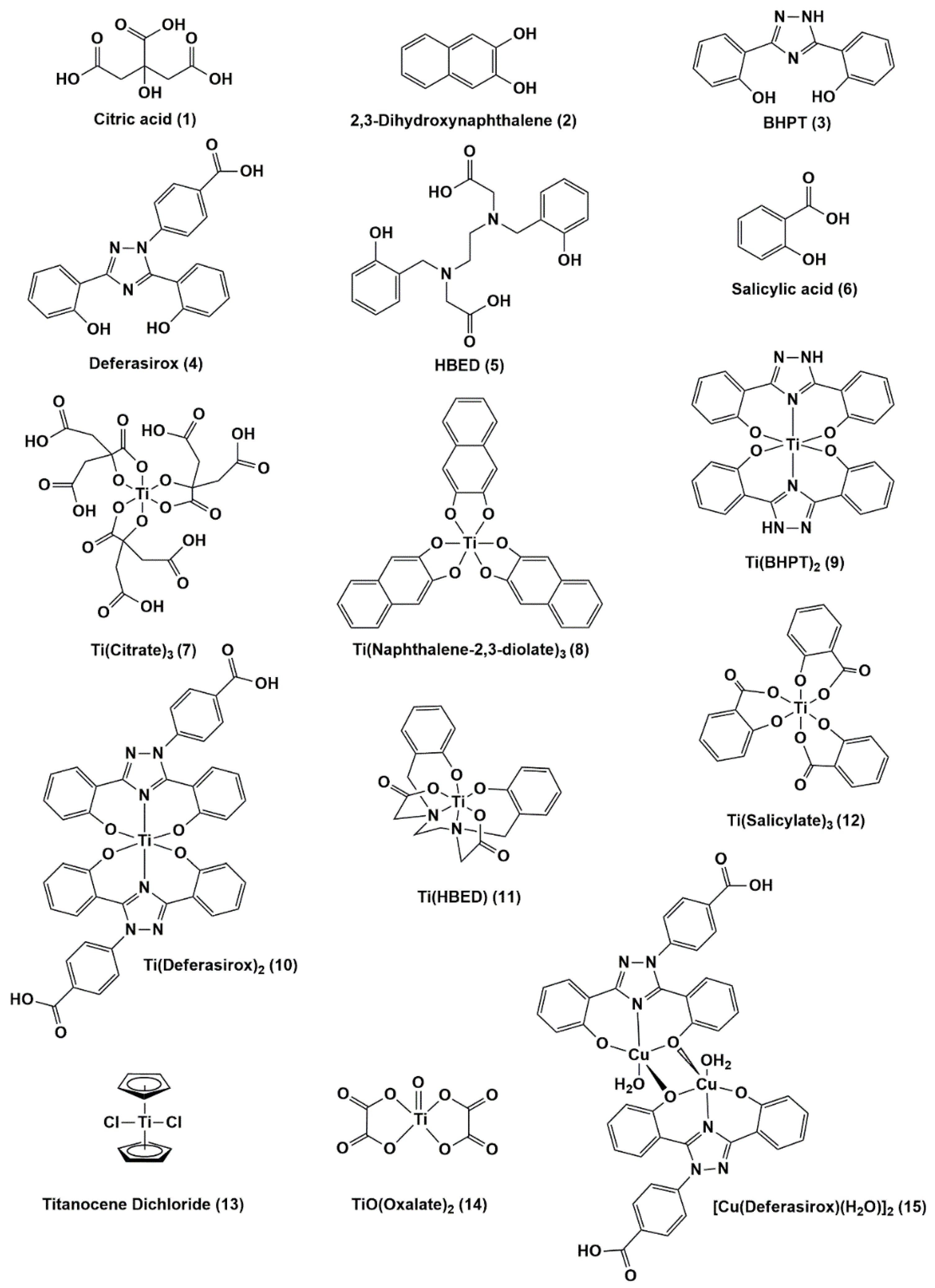
Antibiotics | Free Full-Text | Exploring Titanium(IV) Complexes as Potential Antimicrobial Compounds

Titanium transition metal Chemistry titanium(II) Ti2+ titanium(III) Ti3+ titanium(IV) TiO2+ complex ions TiO2 oxide redox chemical reactions principal oxidation states +2 +3 +4 extraction GCE AS A2 IB A level inorganic chemistry

Effect of the oxidation state of Ti in TMC on catalyst's activity a and... | Download Scientific Diagram
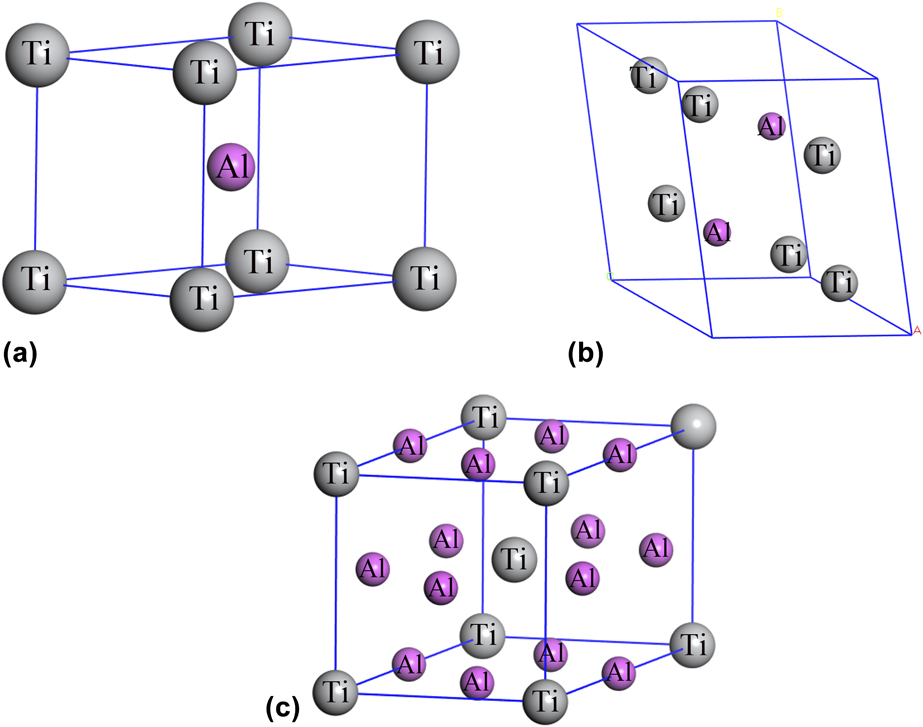
First-principles study on mechanical properties and electronic structures of Ti–Al intermetallic compounds | Journal of Materials Research | Cambridge Core
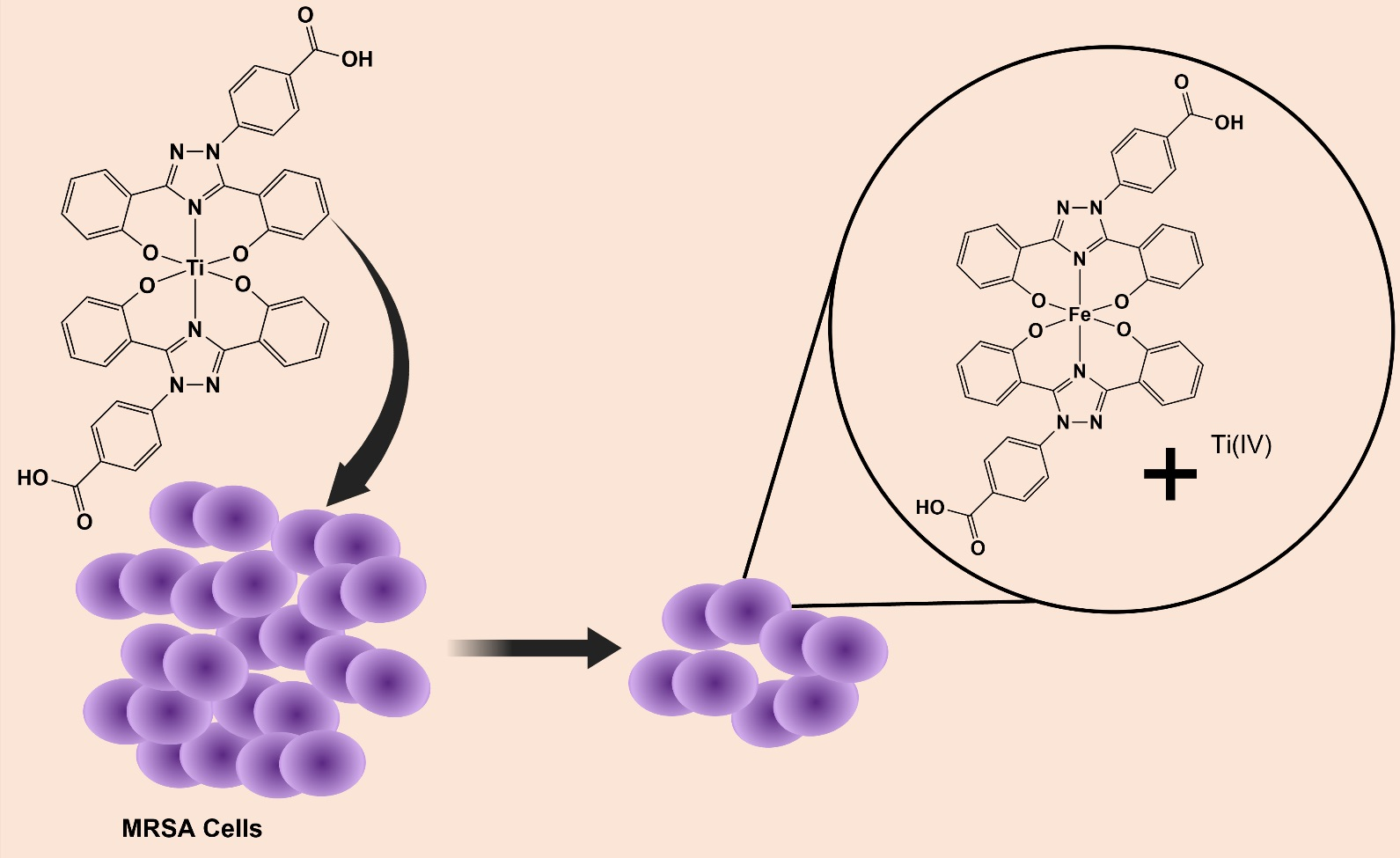
Antibiotics | Free Full-Text | Exploring Titanium(IV) Complexes as Potential Antimicrobial Compounds

Paramagnetism is a property due to the presence of unpaired electrons. In case of transition metals, as they contain unpaired contain unpaired electrons in the (n-1) d orbitals , most of the
![The enthalpy of compound formation in systems Ti-B (curve 1) [1] and... | Download Scientific Diagram The enthalpy of compound formation in systems Ti-B (curve 1) [1] and... | Download Scientific Diagram](https://www.researchgate.net/publication/283657969/figure/fig1/AS:730625522798598@1551206107877/The-enthalpy-of-compound-formation-in-systems-Ti-B-curve-1-1-and-Ti-C-curve-2-2_Q640.jpg)



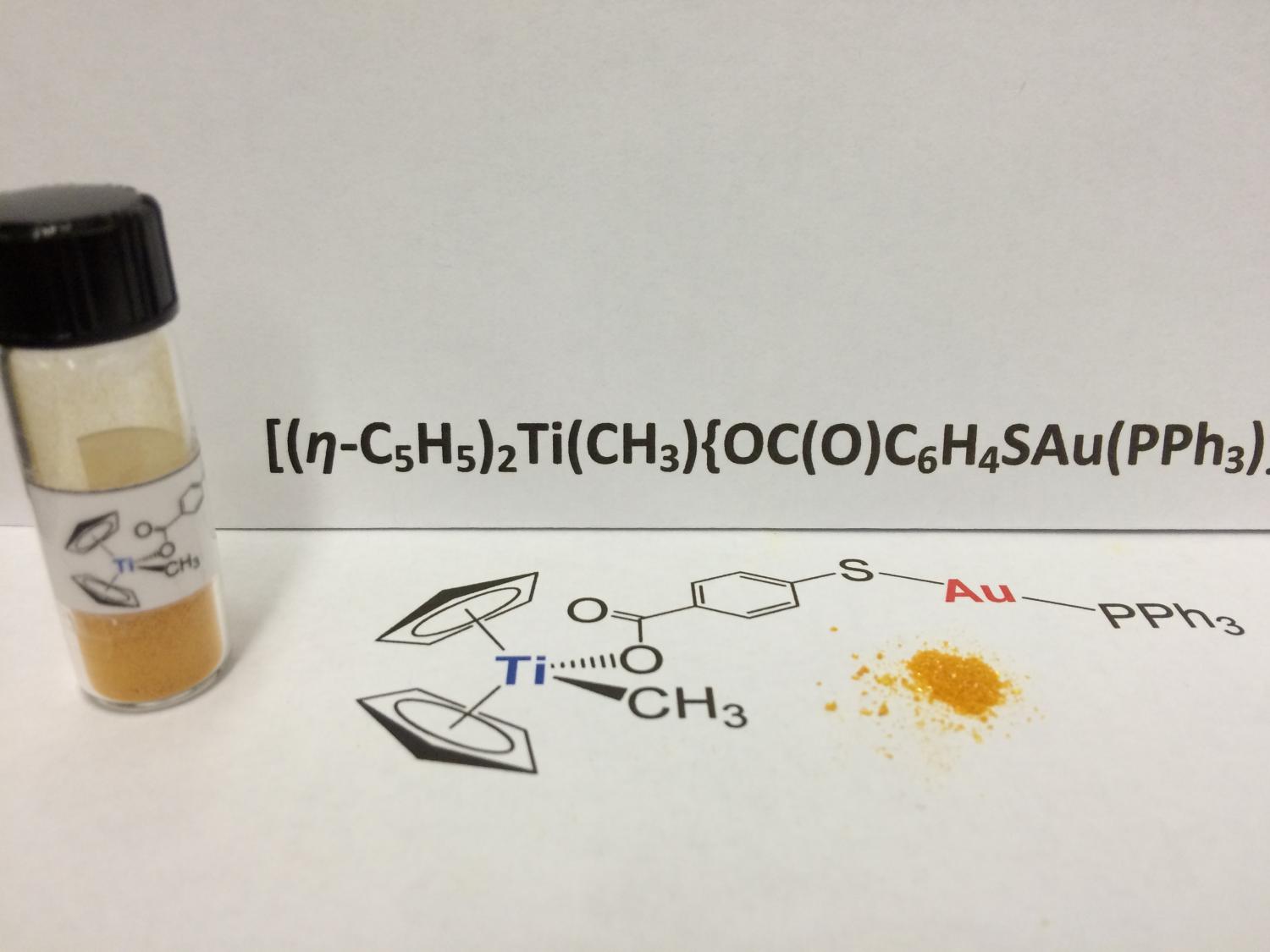



![Ti (Titanium) Compounds [Chemical Structural Class] | TCI AMERICA Ti (Titanium) Compounds [Chemical Structural Class] | TCI AMERICA](https://www.tcichemicals.com/medias/A5020.jpg?context=bWFzdGVyfHJvb3R8NDY1NjB8aW1hZ2UvanBlZ3xoODMvaDlmLzg5Mjg1MTI2MzkwMDYvQTUwMjAuanBnfDYwMWU0MDk2ZWQzMzIxNDRhNzVjMzdiYzJiMmY4ZDdhZDM5MTliNzUzZThhNjc2MDI3YjRhZjViMDFhNWJmNTU)