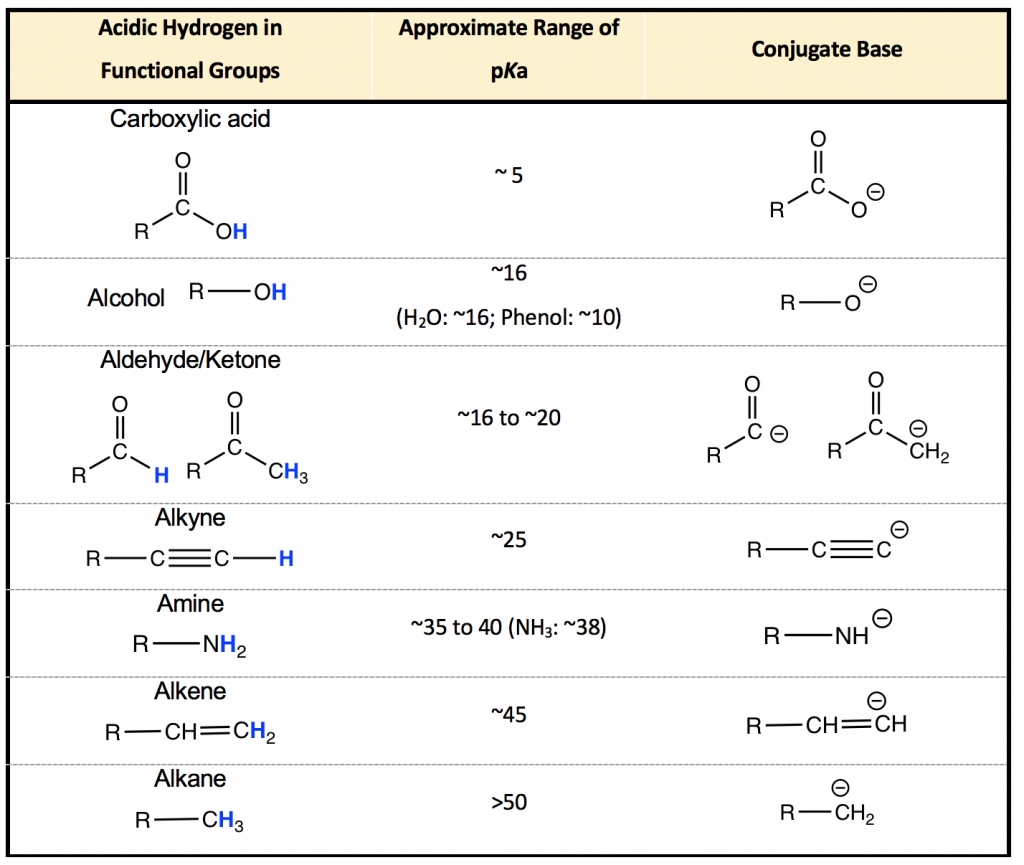
3.3 pKa of Organic Acids and Application of pKa to Predict Acid-Base Reaction Outcome – Organic Chemistry I

The pK(a) value of acetic acid is 4.7447 at 25^(@)C. How would you obtain a buffer of acetic acid and sodium acetate with pH = 4?

pKa value of acetic acid is 4.75. If the buffer solutions contains 0.125 M acetic acid and 0,25 M sodium acetate the pH of buffer solution is :
A hydrogen electrode placed in a buffer solution of CH3COOH and acetic acid in the ratio's x:y and y:x has electrode potential values E1 and E2 volts respectively at 25. the pKa

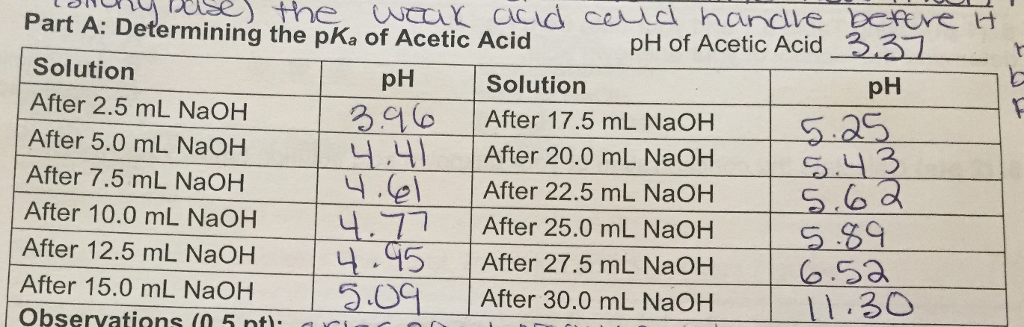
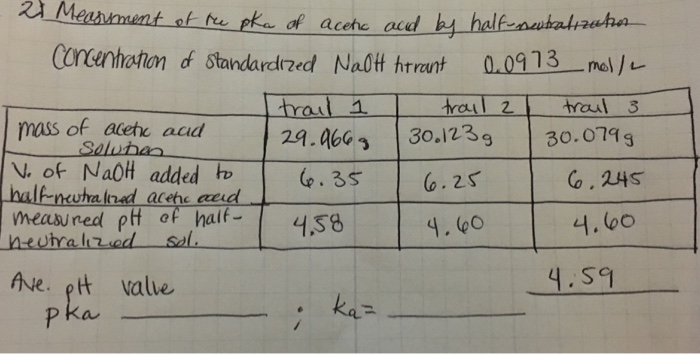
SOLVED: Report Sheet Determination of the Ka of a Weak Acid Name NaOH Molarity IM Acetic Acid Literature value for Ka of Acetic Acid Literature value for pKa of Acetic Acid LkxD -
The pH of an acetic acid solution is 3.26. What is the concentration of acetic acid and what is the percent of acid that's ionized? - Quora
The pKa value of acetic acid is 4.7447 at 25°C. How would you obtain a buffer of acetic acid and sodium acetate with pH = 4? - Sarthaks eConnect | Largest Online Education Community

pKa values in the undergraduate curriculum: introducing pKa values measured in DMSO to illustrate solvent effects | SpringerLink

The pKa of acetic acid and pKb of ammonium hydroxide are 4·76 and 4·75 respectively. Calculate the pH of ammonium acetate solution. from Chemistry Equilibrium Class 11 Nagaland Board

pH calculations and more in fundamentals of pharmaceutics. : Calculate pH of 100 ml buffer solution containing 0.1 g acetic acid and 0.2 g sodium actetate.

OneClass: What concentrations of acetic acid (pKa = 4.76) and acetate would be required to prepare a ...
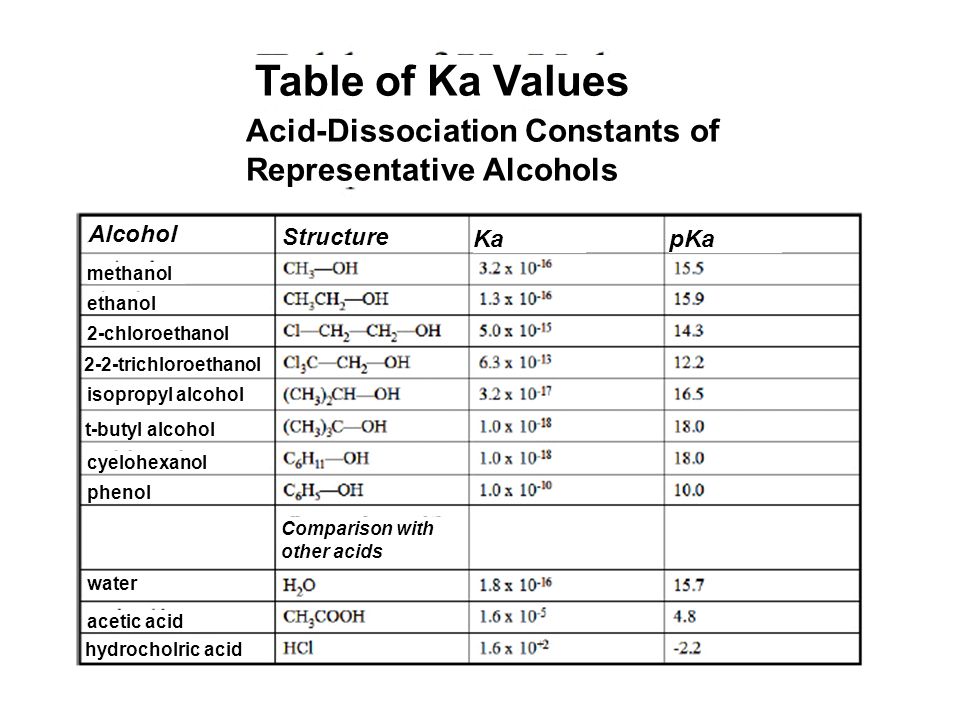
Table of Ka Values Acid-Dissociation Constants of Representative Alcohols Alcohol Structure Ka pKa methanol ethanol 2-chloroethanol 2-2-trichloroethanol. - ppt video online download
![SOLVED: [ ] pKa of weak acids at 25*C Name Formula acetic acid CH3COzH benzoic acid C6HsCOzH butanoic acid CzHzCOzH 4-chlorobutanoic acid CzHsCICOzH crotonic acid CzHsCOzH oxalic acid HzC204 phosphoric acid HaPOa SOLVED: [ ] pKa of weak acids at 25*C Name Formula acetic acid CH3COzH benzoic acid C6HsCOzH butanoic acid CzHzCOzH 4-chlorobutanoic acid CzHsCICOzH crotonic acid CzHsCOzH oxalic acid HzC204 phosphoric acid HaPOa](https://cdn.numerade.com/ask_images/346c7f228e504822a839e28cc57b4b36.jpg)





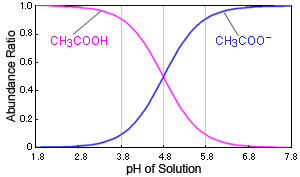

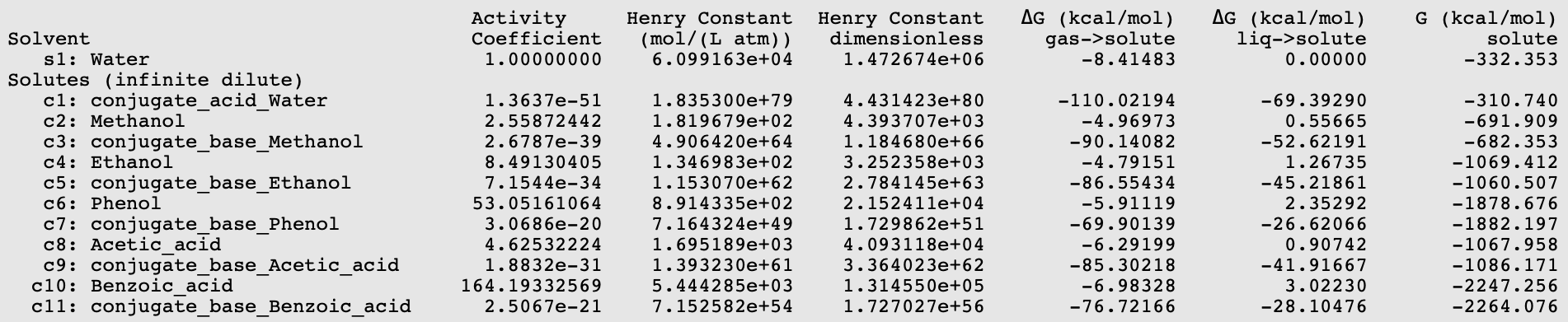

:max_bytes(150000):strip_icc()/what-is-pka-in-chemistry-605521_FINAL2-9fdfc39e9aa34caa96d6e74a2c687707.png)