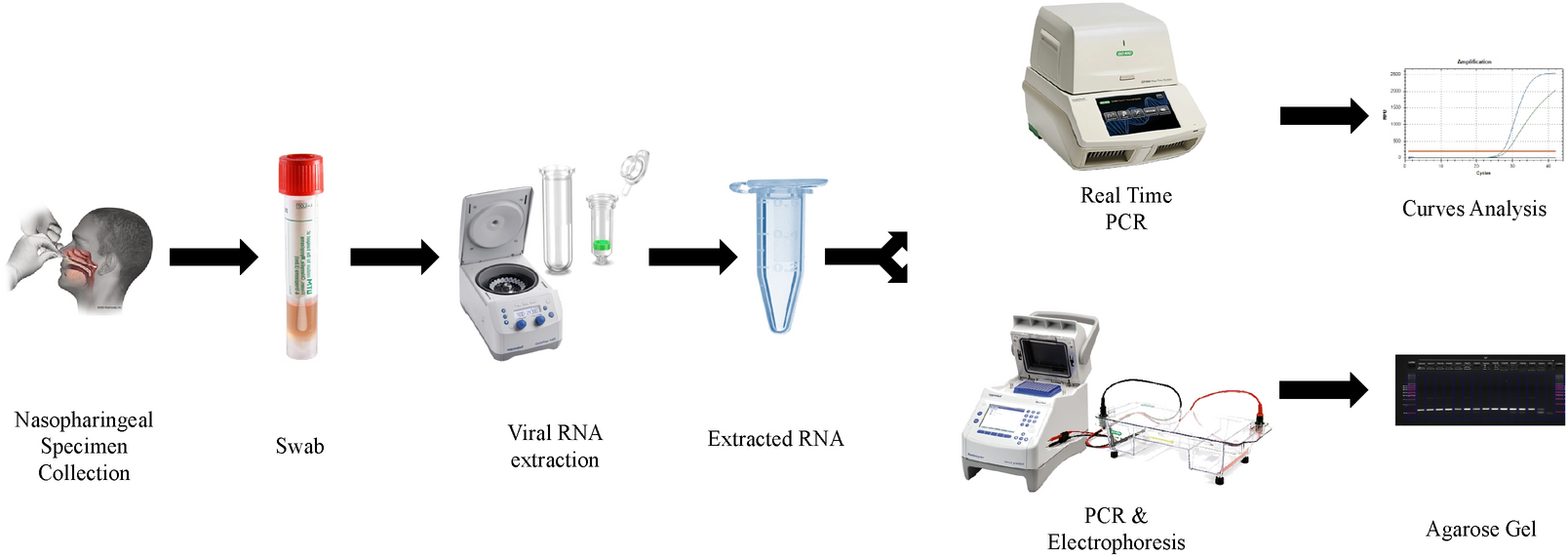
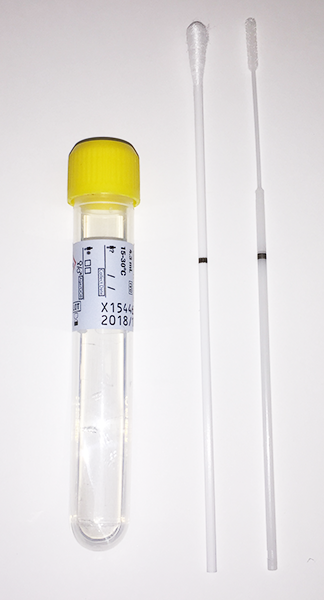
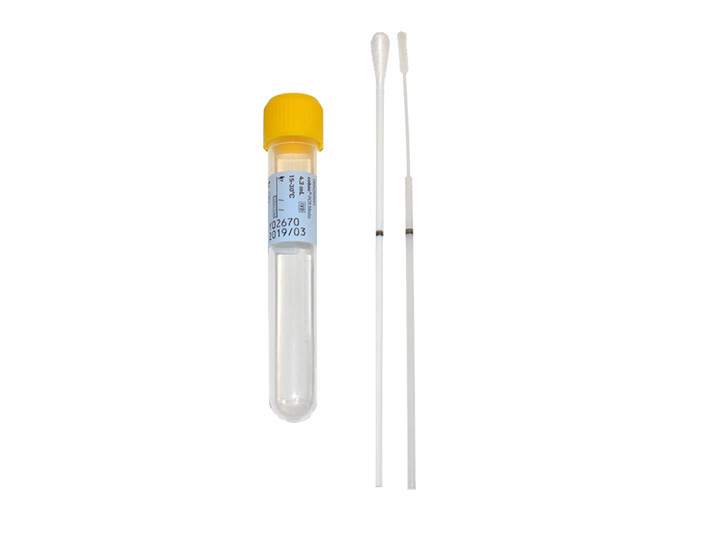
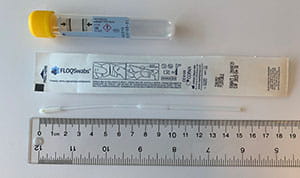
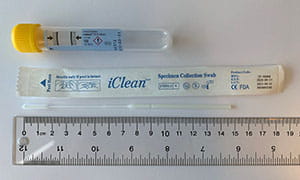
The table below provides guidance on appropriate specimen types based on swab collection kits that may be in your inventory. I
Molecular Testing for Sexually Transmitted Infections: Phase 3 Transition to Roche Method and Collection Devices

Chlamydia trachomatis/Neisseria gonorrhoeae (CT/NG) – Nucleic Acid Amplification Testing (NAAT) | Public Health Ontario