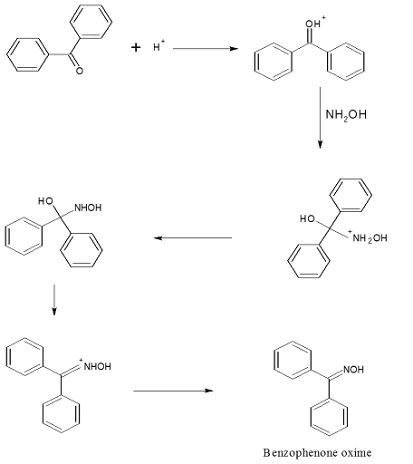
![Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund) Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)](https://acswebcontent.acs.org/prfar/2011/images/Paper_11567_abstract_15187_0.gif)
Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)
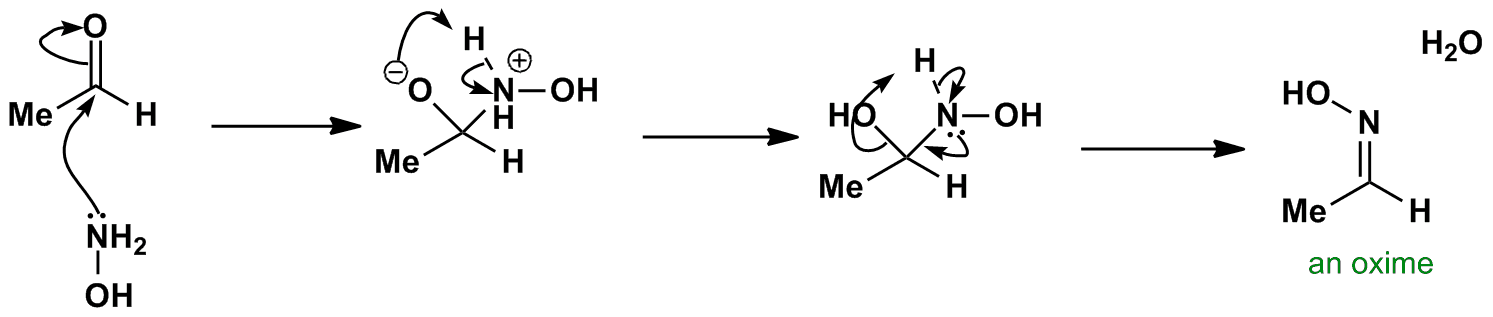
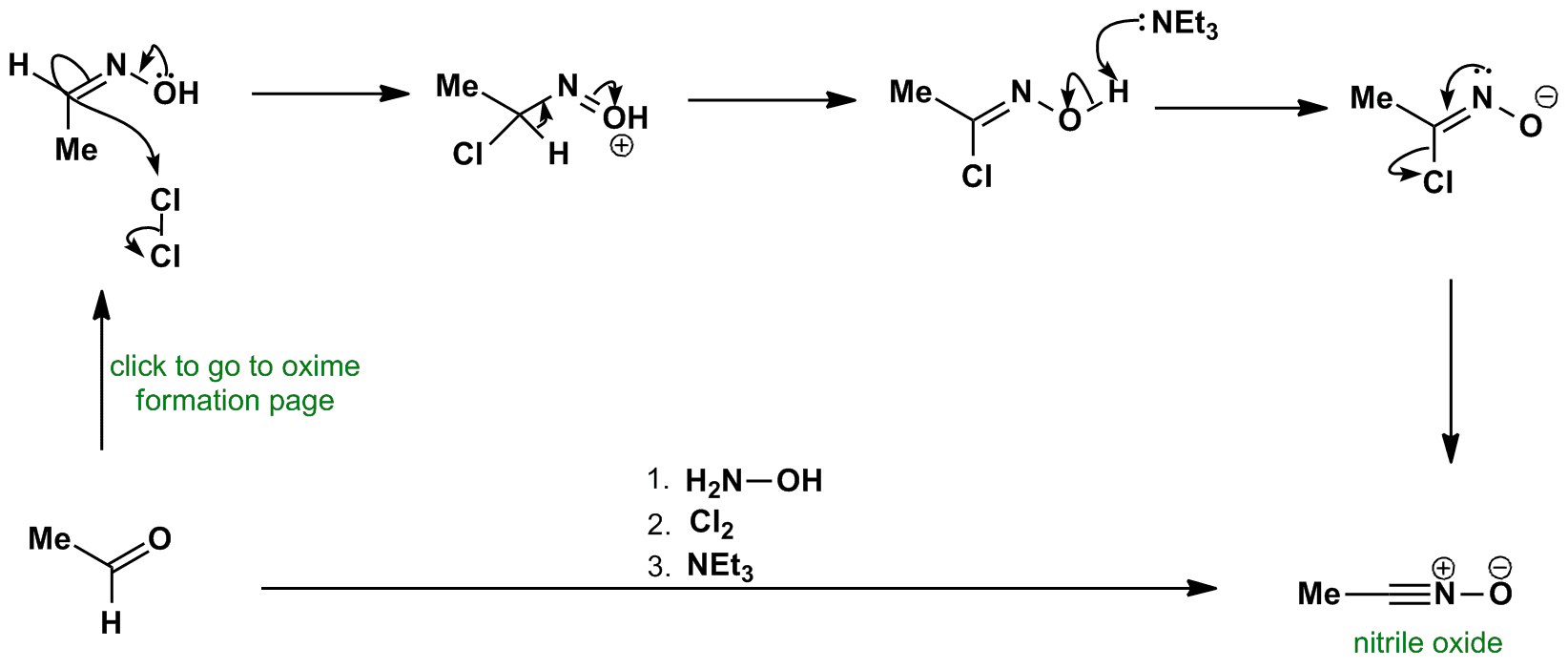
![Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund) Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)](https://acswebcontent.acs.org/prfar/2011/images/Paper_11567_abstract_15186_0.gif)
Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)
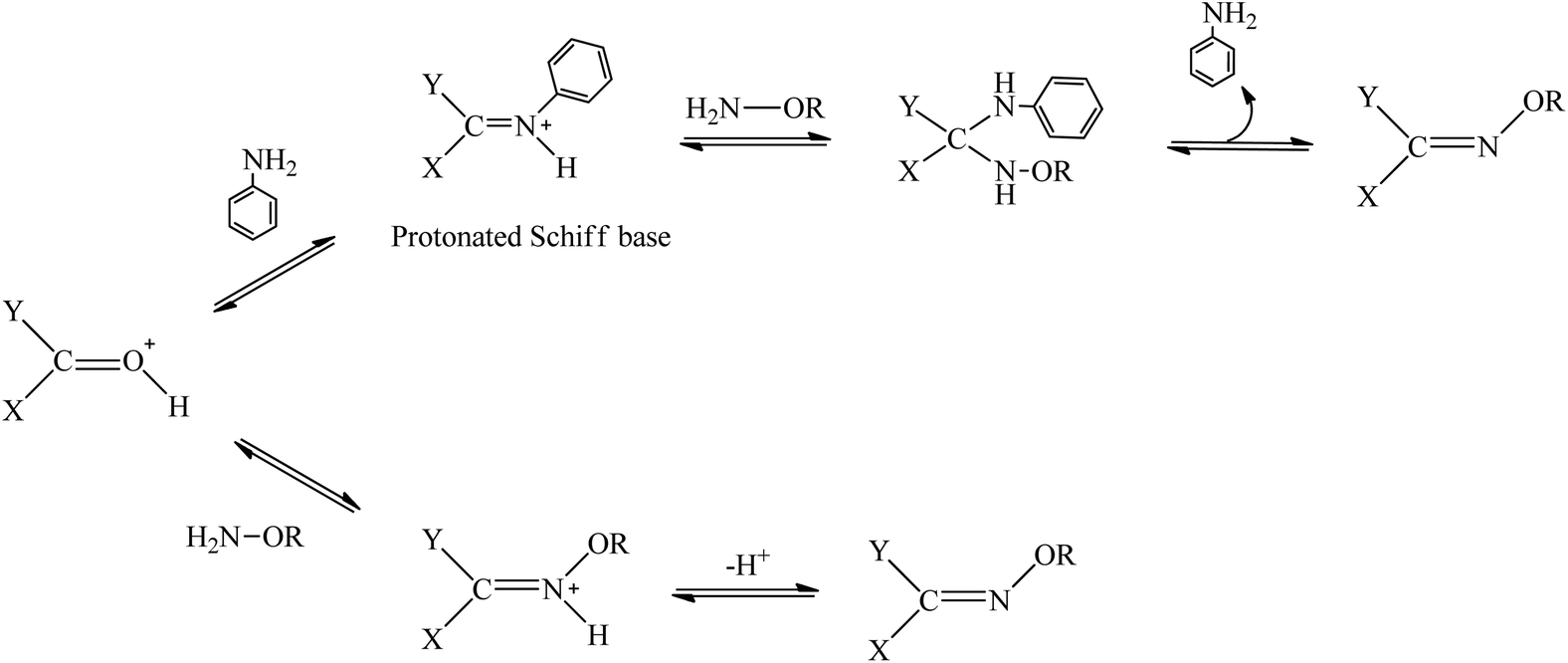
The emergence of oxime click chemistry and its utility in polymer science - Polymer Chemistry (RSC Publishing) DOI:10.1039/C6PY00635C
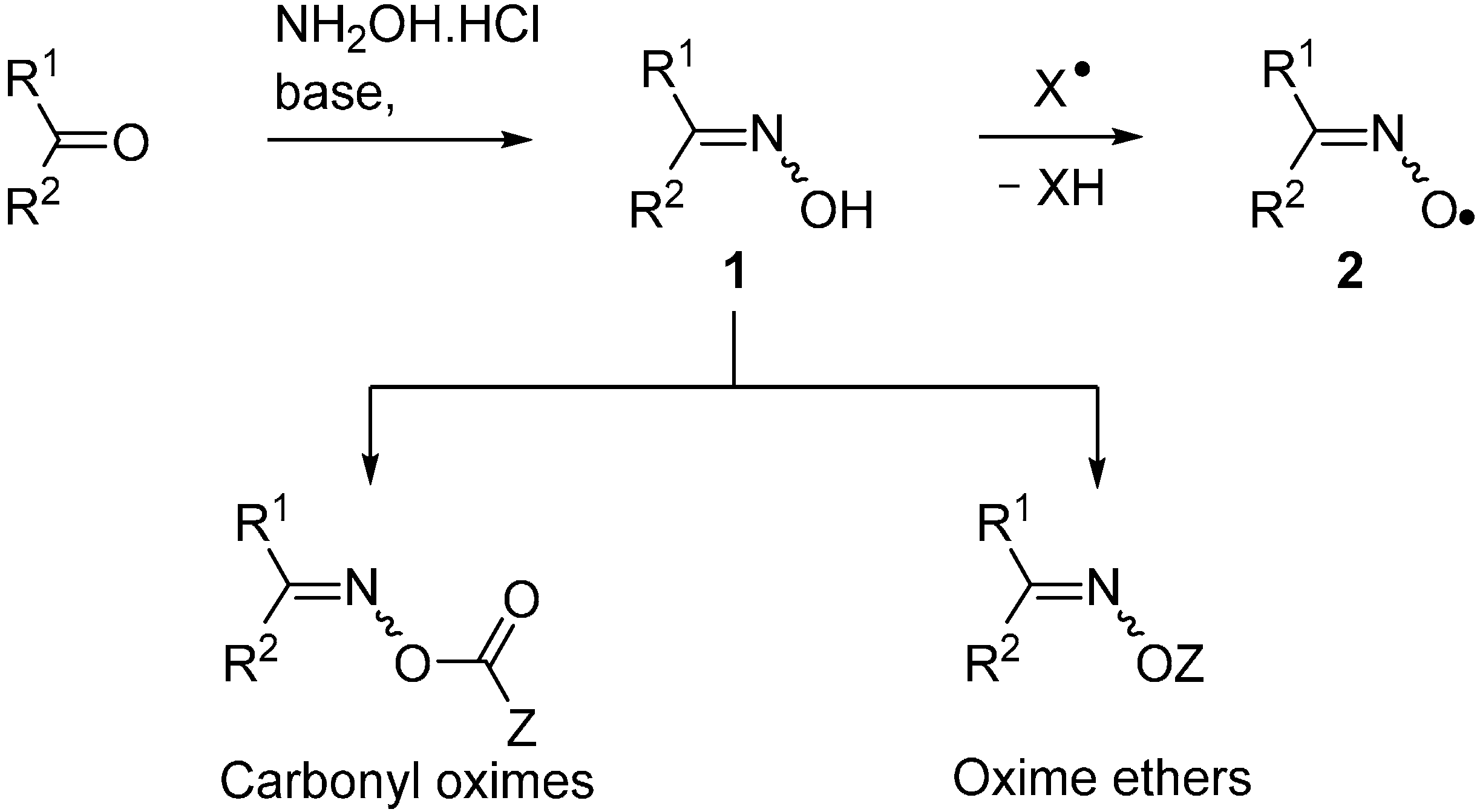
Molecules | Free Full-Text | Functionalised Oximes: Emergent Precursors for Carbon-, Nitrogen- and Oxygen-Centred Radicals

Molecules | Free Full-Text | Amidoximes and Oximes: Synthesis, Structure, and Their Key Role as NO Donors

The local and natural sources in synthetic methods: the practical synthesis of aryl oximes from aryl aldehydes under catalyst-free conditions in mineral water | SpringerLink