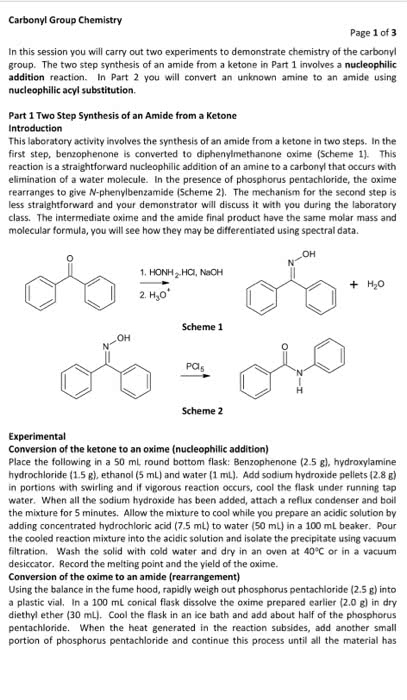
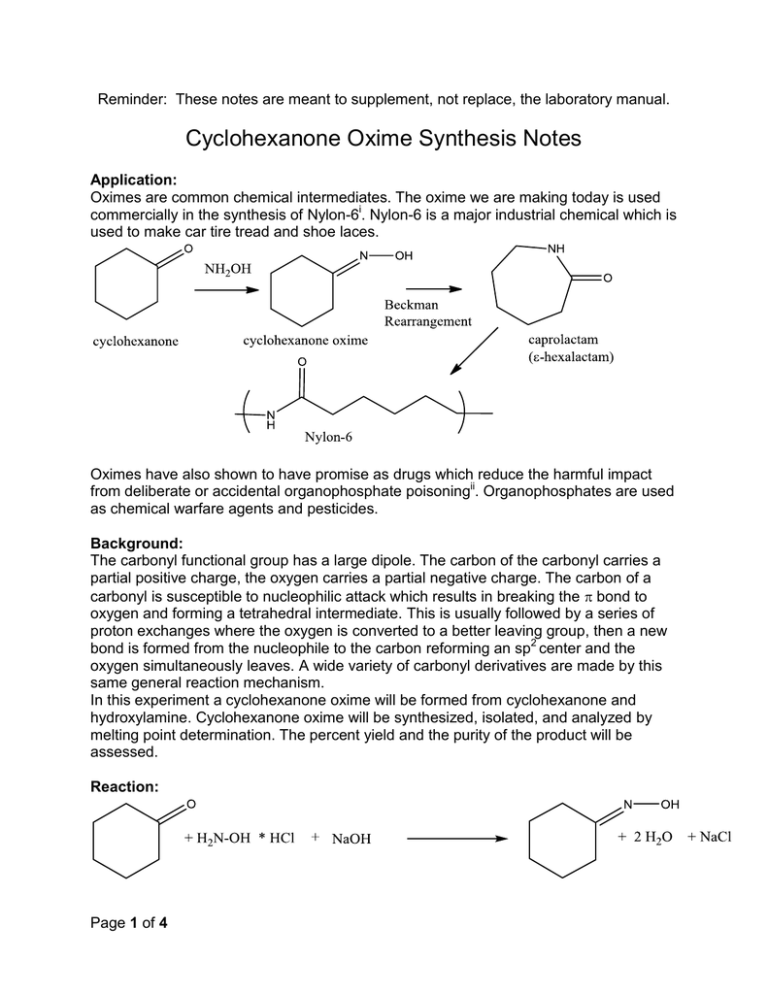
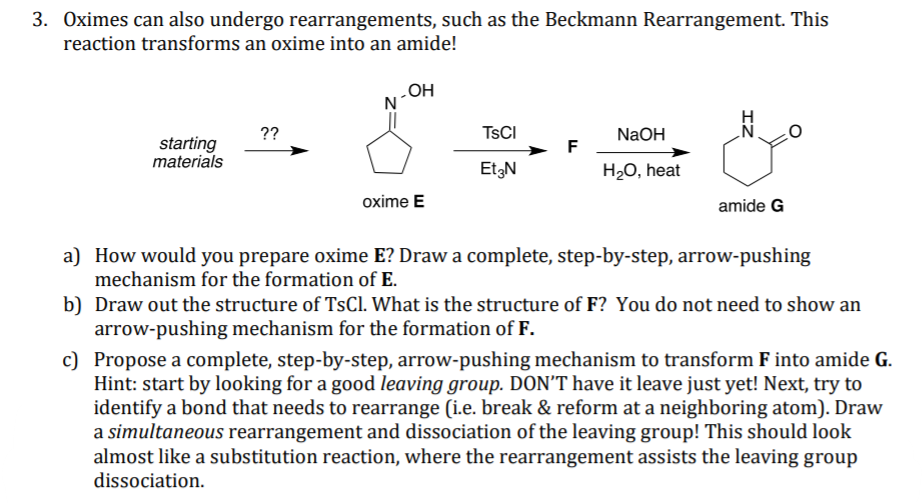
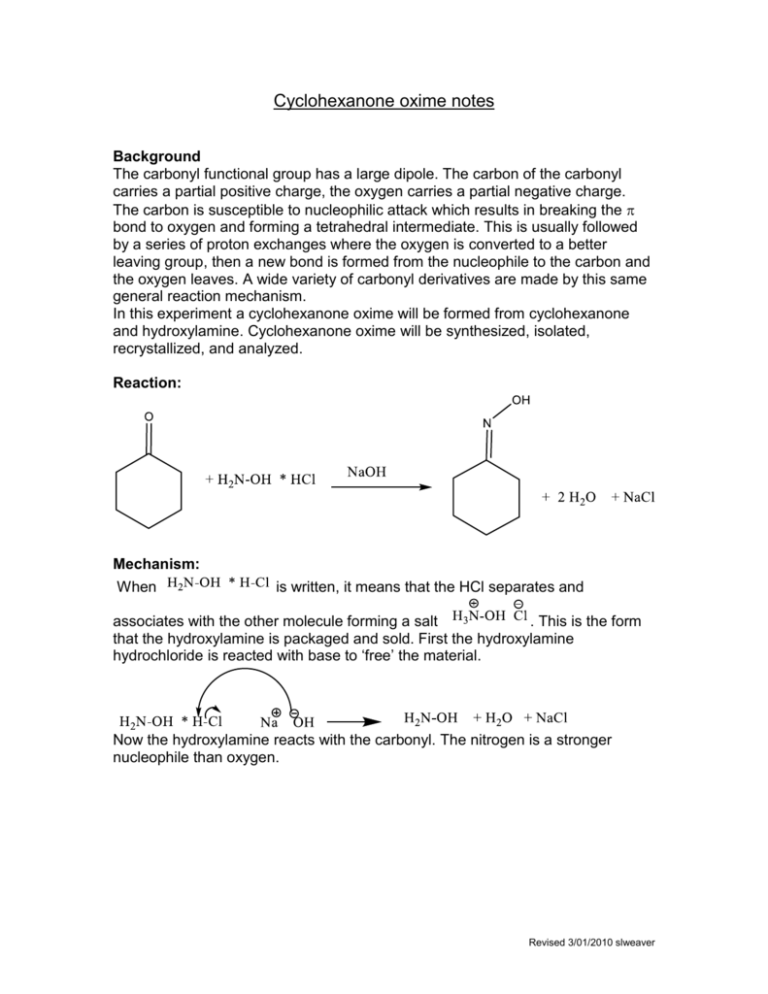
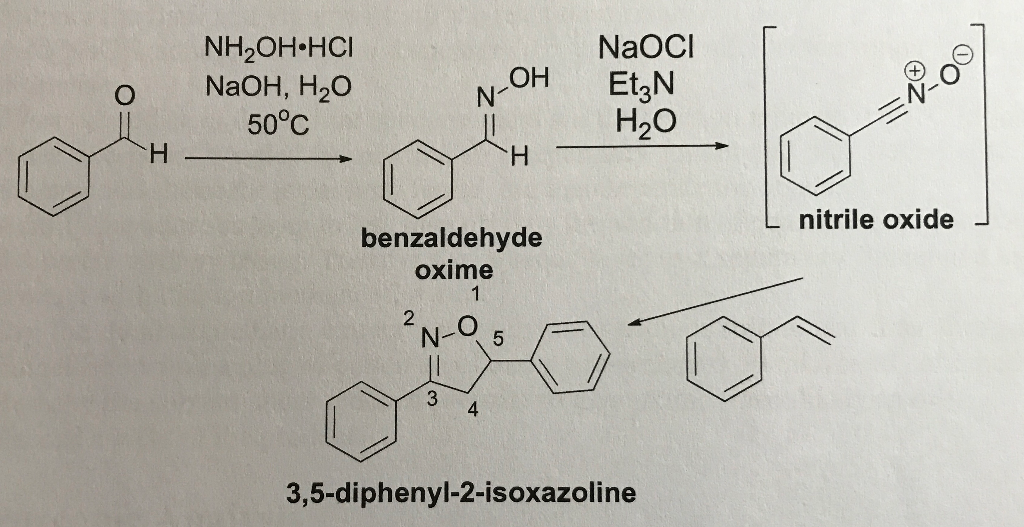
Beckmann rearrangement of cyclohexanone oxime in a microchemical system: The role of SO3 and product inhibition - Zhang - 2012 - AIChE Journal - Wiley Online Library
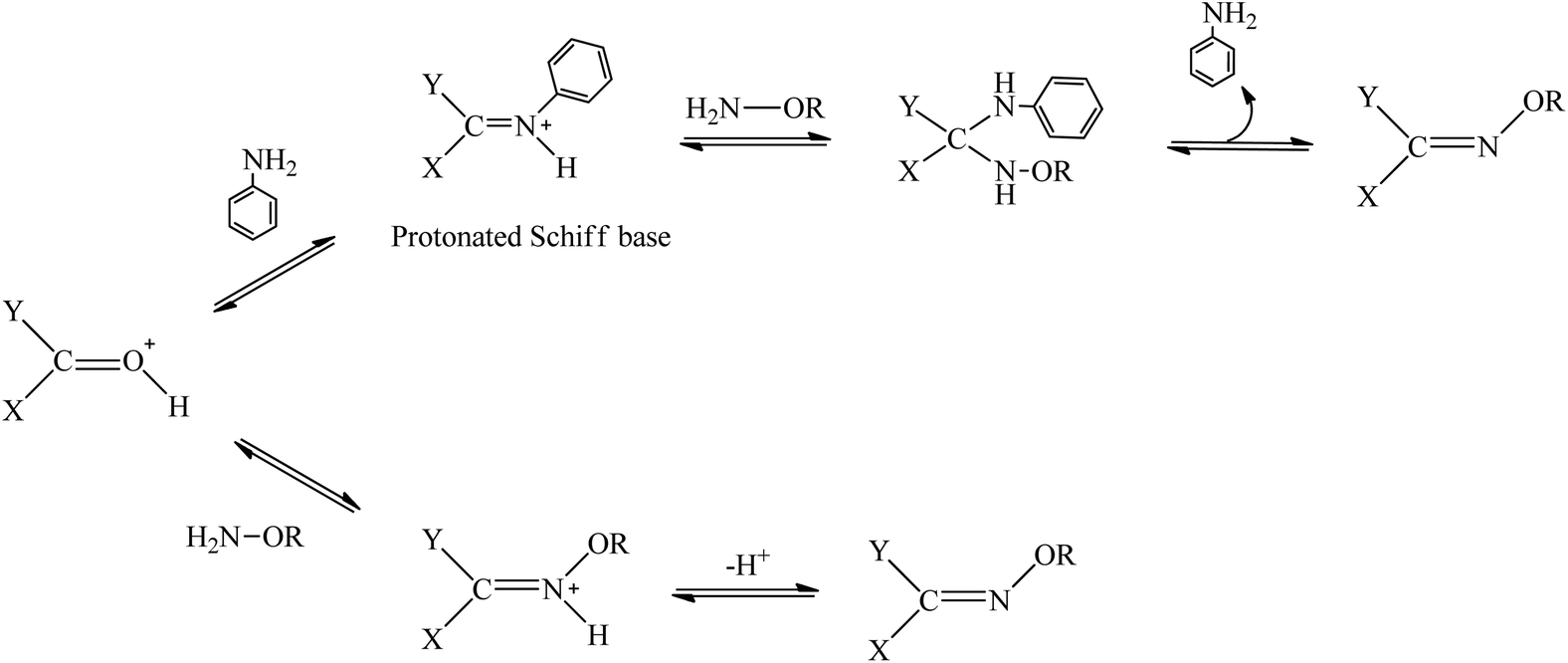
The emergence of oxime click chemistry and its utility in polymer science - Polymer Chemistry (RSC Publishing) DOI:10.1039/C6PY00635C

An organic compound (A) (C(8)H(14)O) forms an oxime and gives a positive haloform reaction. On ozonolysis, it gives acetone and a compound (B) (C(5)H(8)O(2)) . (B) forms a dioxime and on subjecting
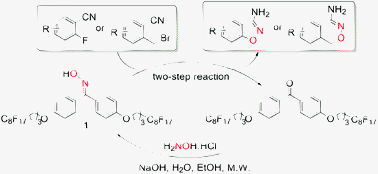
Application of a recyclable fluorous oxime in the convenient synthesis of 3-amino-1,2-benzisoxazoles and 4-amino-1H-2,3-benzoxazines - Green Chemistry (RSC Publishing)

SOLVED: Em UcdQ 2 Reaction of unsymmetrical ketones with hydroxylamine gives mixture of oxime somers OH R'N R? NH2e OHHCI NaOH OH and / Or a? Describe how You might distinguish the
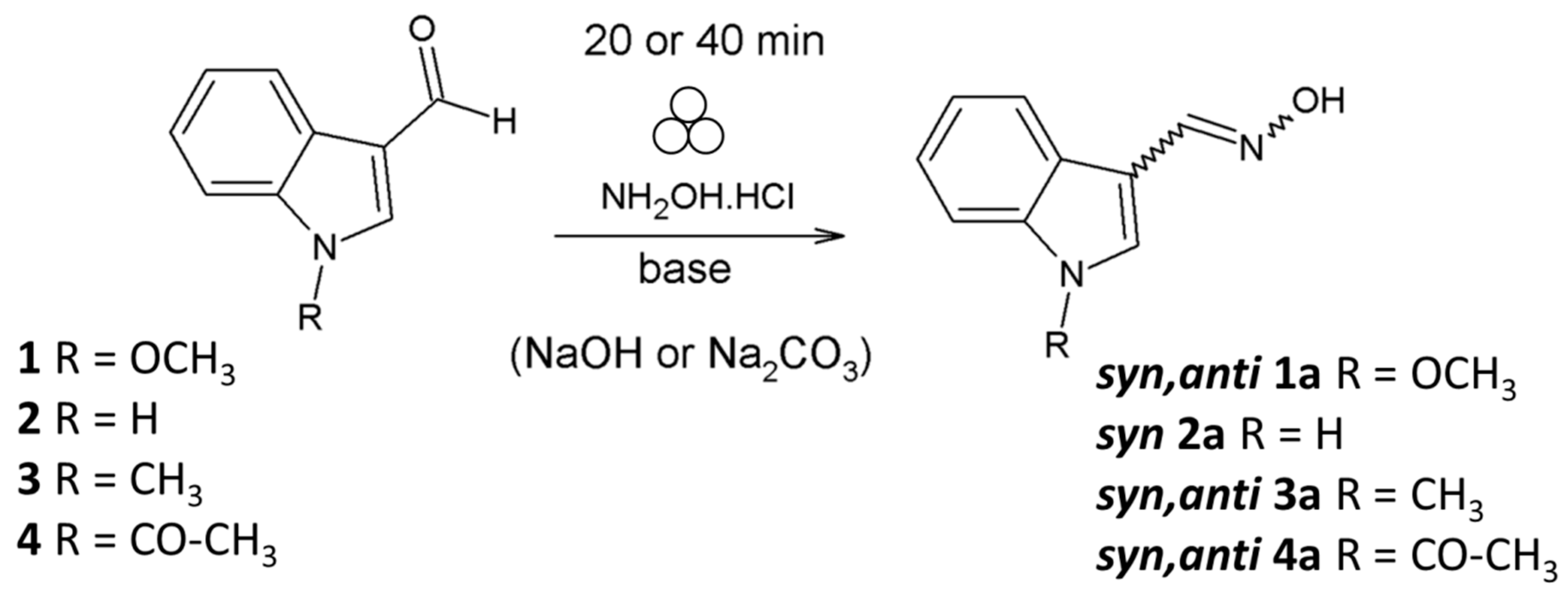
Molecules | Free Full-Text | Mechanochemical Synthesis and Isomerization of N-Substituted Indole-3-carboxaldehyde Oximes †

Compound X `(C_7H_14O_4)` on ozonolysis gives (Y) and (Z).(Z) is the aldehyde which gives only oxime with `NH_2-OH` On treatment with `I_2/(NaOH)`.(Y) - Sarthaks eConnect | Largest Online Education Community
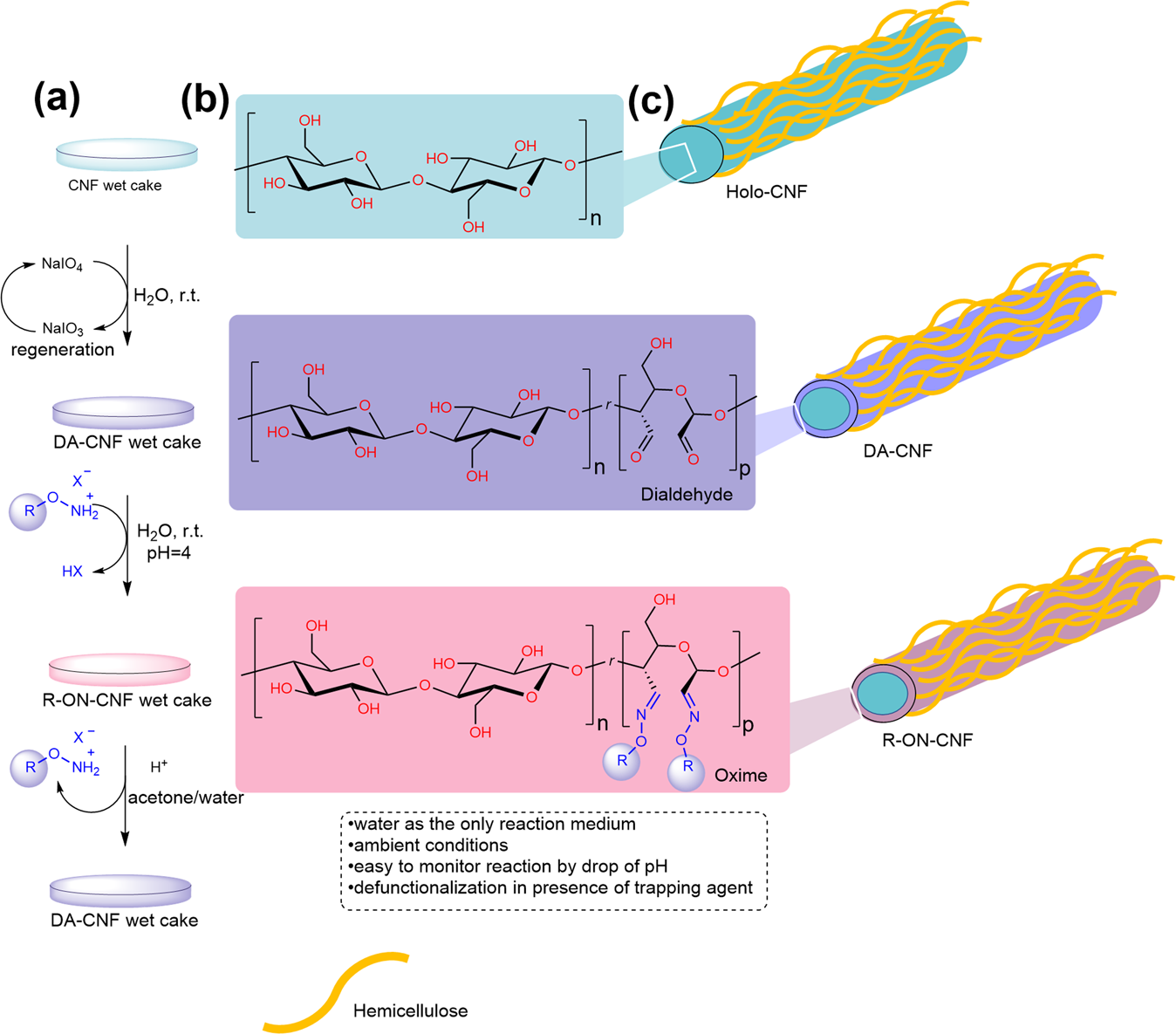
Aqueous synthesis of highly functional, hydrophobic, and chemically recyclable cellulose nanomaterials through oxime ligation | Nature Communications