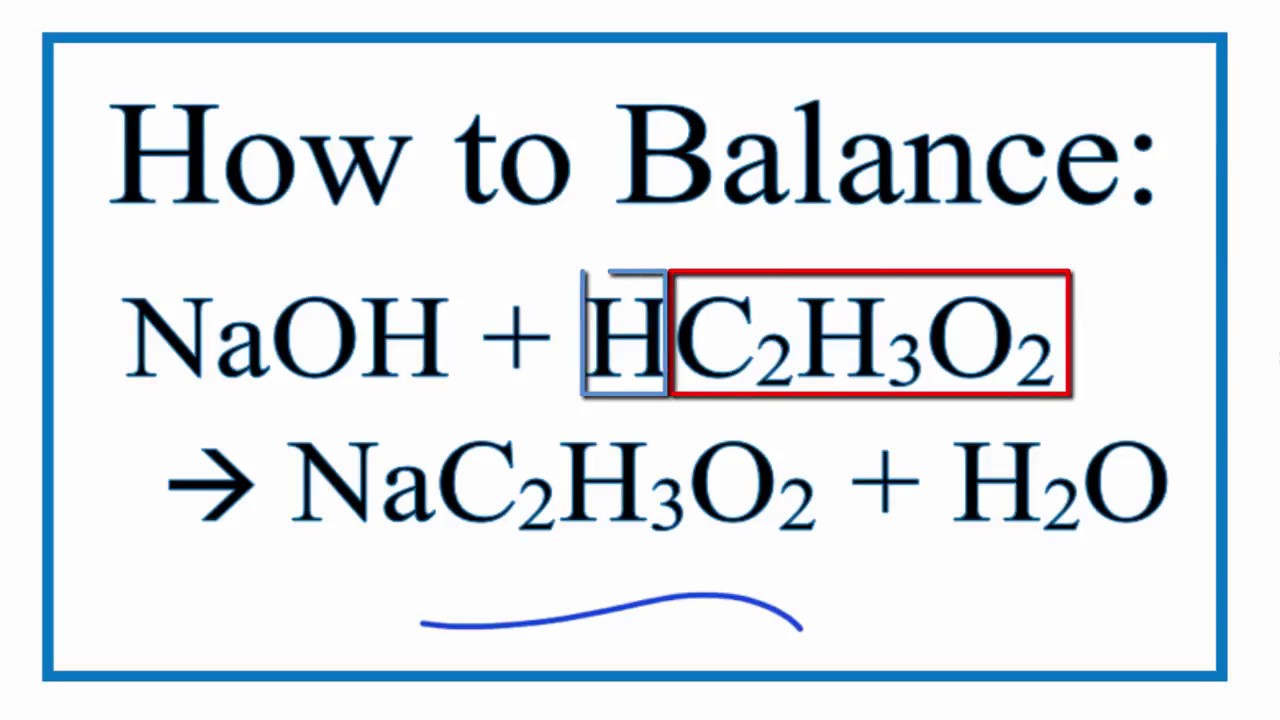
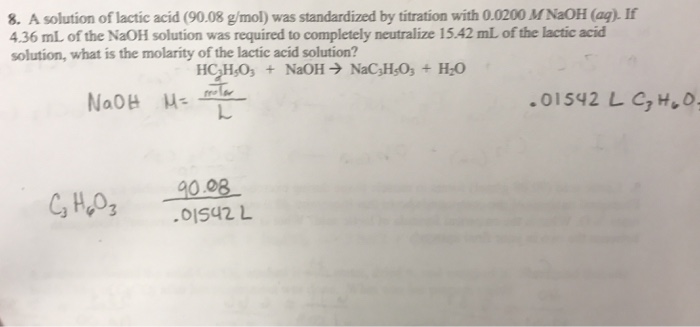OneClass: 3. (15 pts) How many grams of lactic acid, CH3CHOHCOOH, togetherwith 2.00 grams of NaOH, mu...

pH titration curves of free lactic acid and Cr 3þ /lactic acid in 1 :... | Download Scientific Diagram

pH titration curve of the aqueous lactic acid solution. The original... | Download Scientific Diagram

Selective Conversion of Glucose into Lactic Acid with Transition Metal Ions in Diluted Aqueous NaOH Solution | ACS Sustainable Chemistry & Engineering
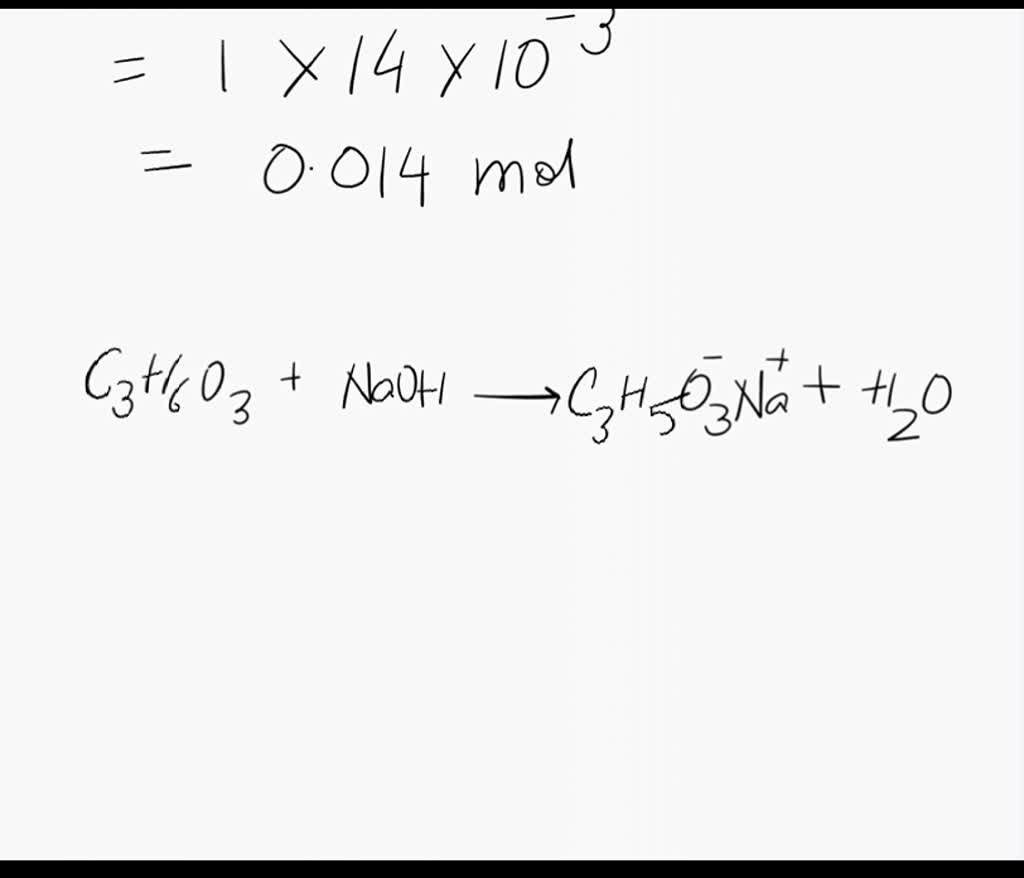
SOLVED: Note: Problem #Lis required Lactic acid builds up in the muscles when one exercises. 200 mL of 0.400 M lactic acid (HLac) was reacted with 100 mL of 0.500 M NaOH

SOLVED: Question 14 1pts 100 mLof 0.1 M NaOH is added to 55 mL of 0.2 M lactic acid. The pKa of lactic acid is 4.1. The resulting mixture has a pH close to: 6.9 0 5.1 0 8.3 0 44 Question 15

SOLVED: Question The reaction of lactic acid with sodium hydroxide proceeds by the following equation (Let HA represent lactic acid). HA + NaOH - NaA + H,O The end point In a

Lecture 183/1/06. What is a buffer? pH of a buffer system What is the pH of a buffer that is 0.12 M lactic acid HC 3 H 5 O 3 and 0.1 M sodium lactate? - ppt download

SOLVED:Lactic acid, C3 H6 O3, is the acid present in sour milk. A 0.100 -g sample of pure lactic acid requires 12.95 mL of 0.0857 M sodium hydroxide for complete reaction. How

If you wanted to hydrolyze 1 mole of PLA into 1 mole of sodium lactic acid, how many moles of sodium hydroxide would you need? | Homework.Study.com

pH titration curve of the aqueous lactic acid solution. The original... | Download Scientific Diagram
![SOLVED:How many microliters of 1.000 M NaOH solution must be added to 25.00 mL of a 0.1000 M solution of lactic acid [CH3 CH(OH) COOH. or .HC3 H5 O3] to produce a SOLVED:How many microliters of 1.000 M NaOH solution must be added to 25.00 mL of a 0.1000 M solution of lactic acid [CH3 CH(OH) COOH. or .HC3 H5 O3] to produce a](https://cdn.numerade.com/previews/a71cd3ce-5045-4f39-a5f1-44cb28587742.gif)
SOLVED:How many microliters of 1.000 M NaOH solution must be added to 25.00 mL of a 0.1000 M solution of lactic acid [CH3 CH(OH) COOH. or .HC3 H5 O3] to produce a

Hydrothermal conversion of glycerol to lactic acid catalyzed by Cu/hydroxyapatite, Cu/MgO, and Cu/ZrO2 and reaction kinetics - ScienceDirect

Scheme 1. Reagents and conditions: (a) lactic acid, 4 M HCl, reflux;... | Download Scientific Diagram

OneClass: Write a balanced equation for the titration of NaOH with: Citric acid (acid in candy) Lacti...