![Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund) Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)](https://acswebcontent.acs.org/prfar/2011/images/Paper_11567_abstract_15186_0.gif)
Report: [3,3]-Rearrangements of O-Vinyl Oximes: Stereoselective Synthesis of 1,4-Dicarbonyl Compounds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)

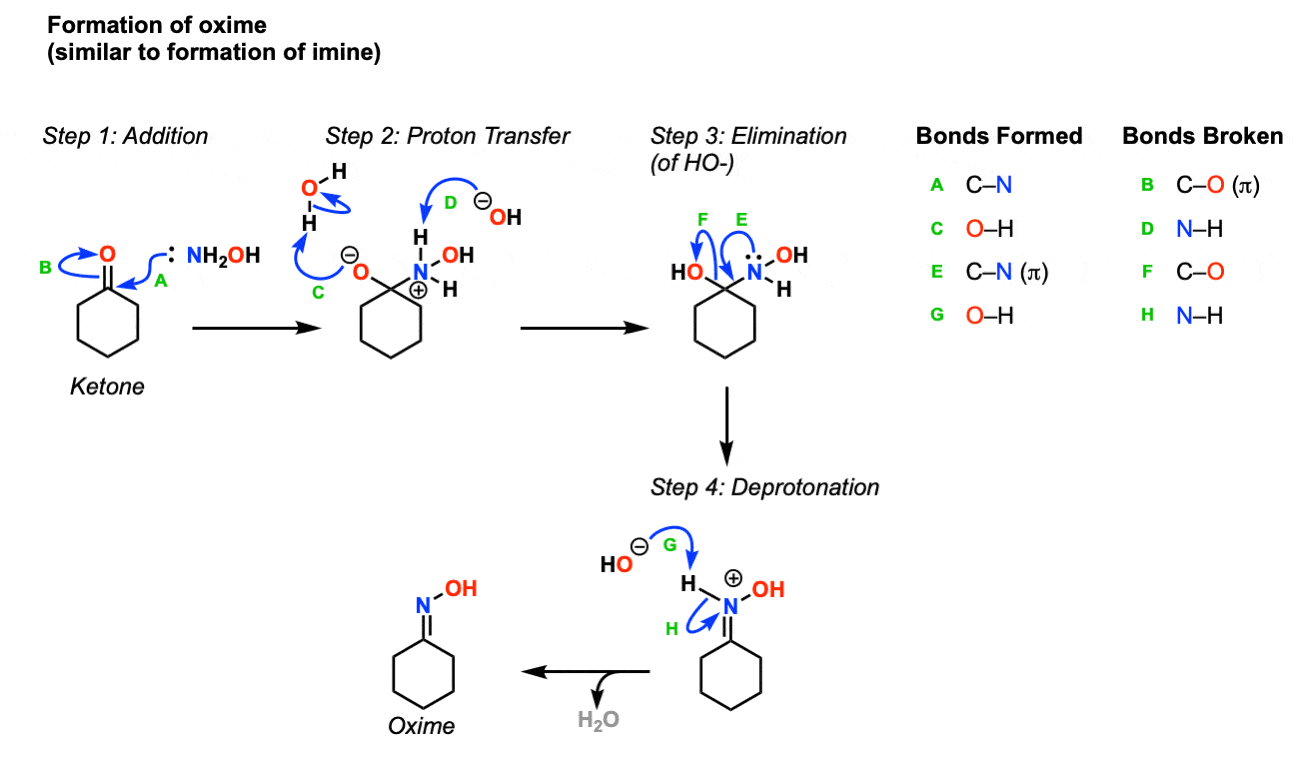
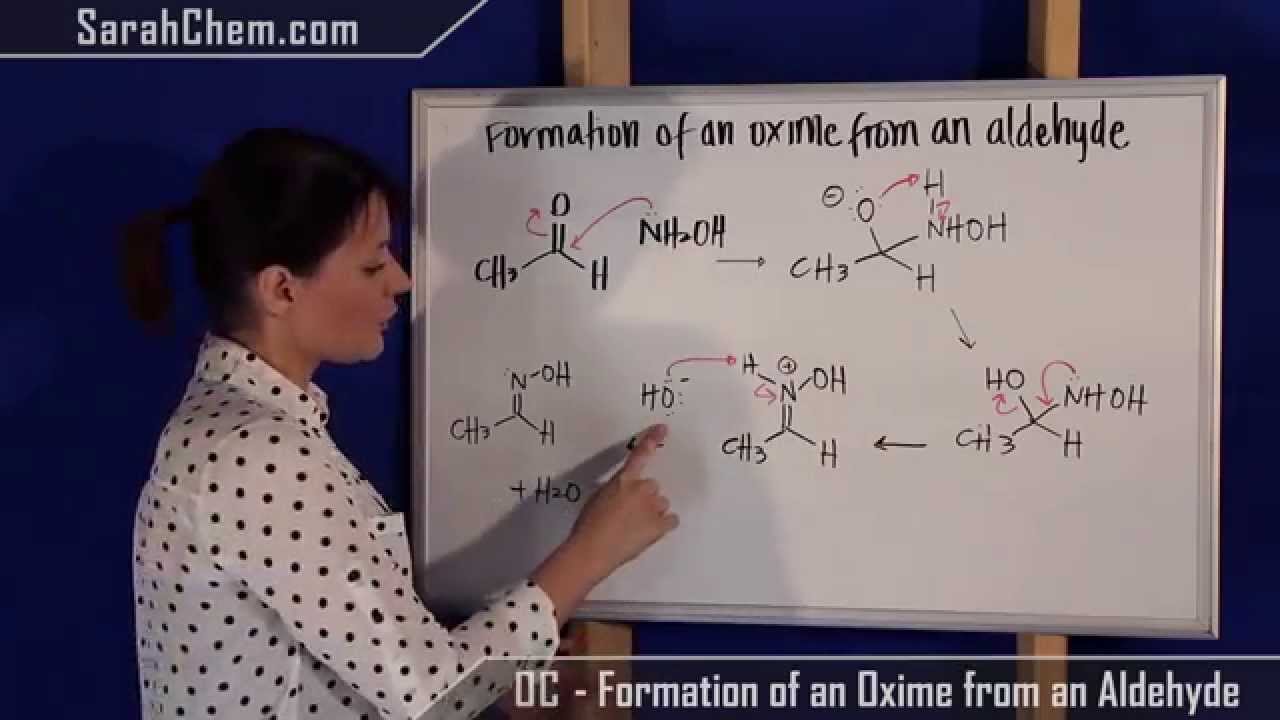
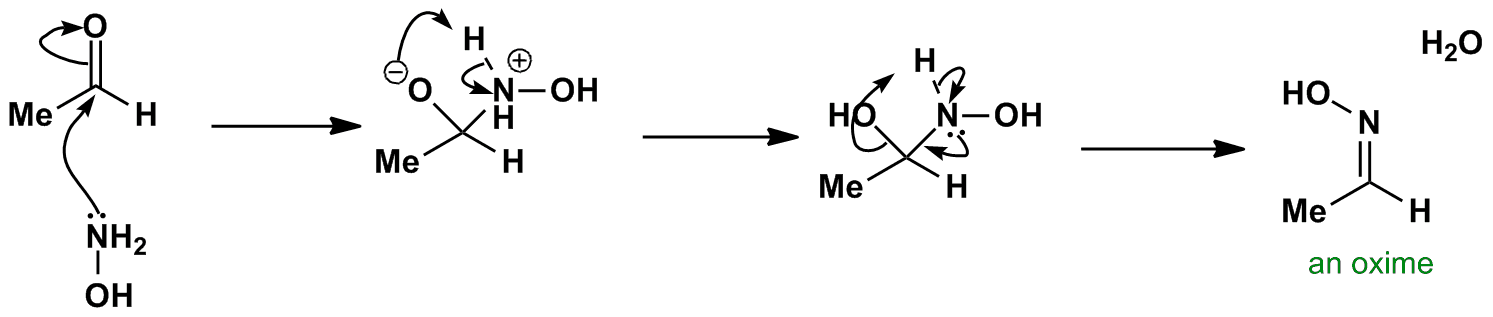
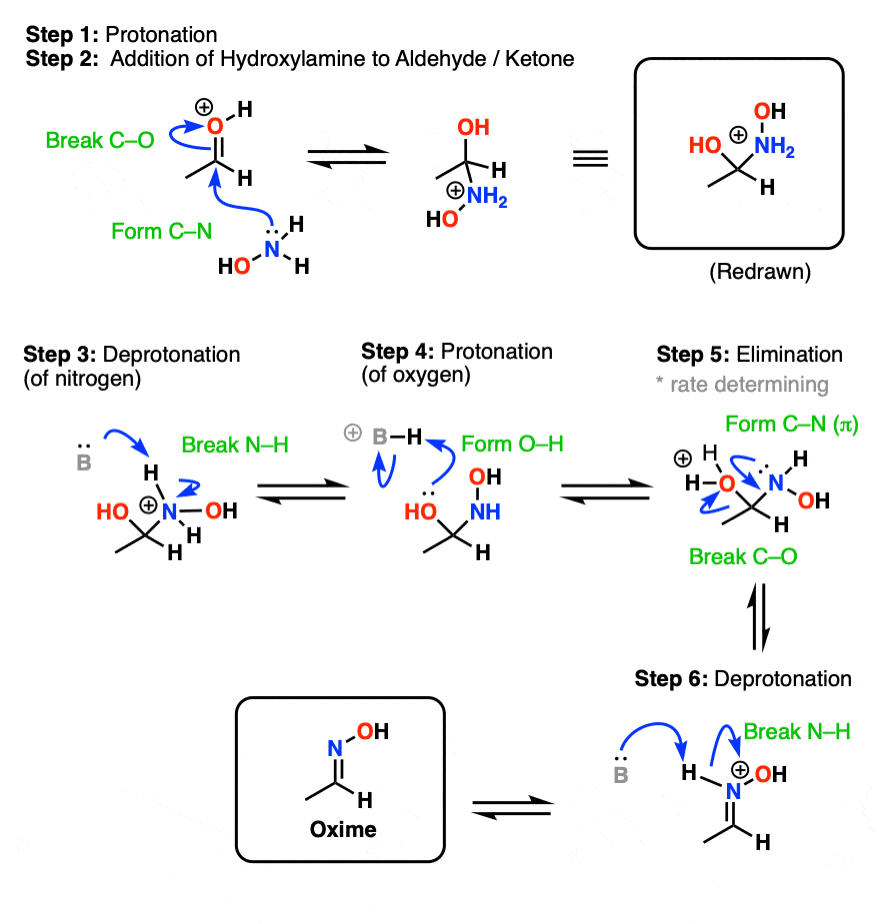
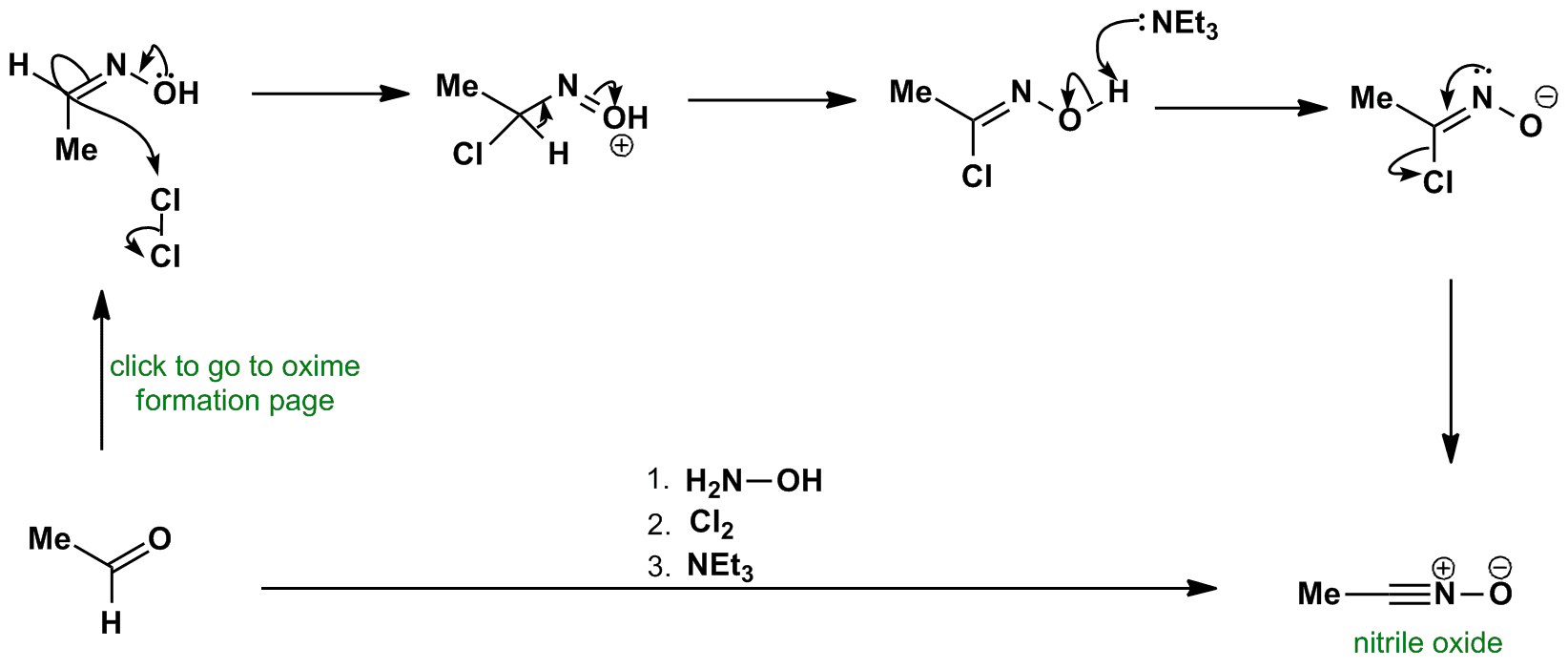
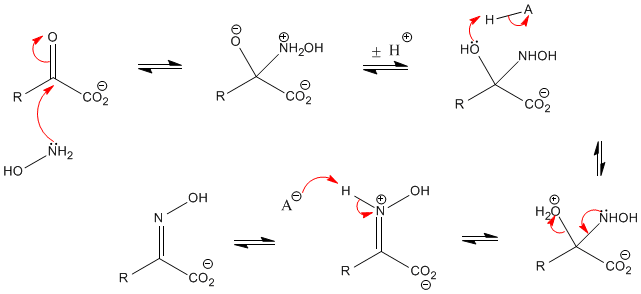
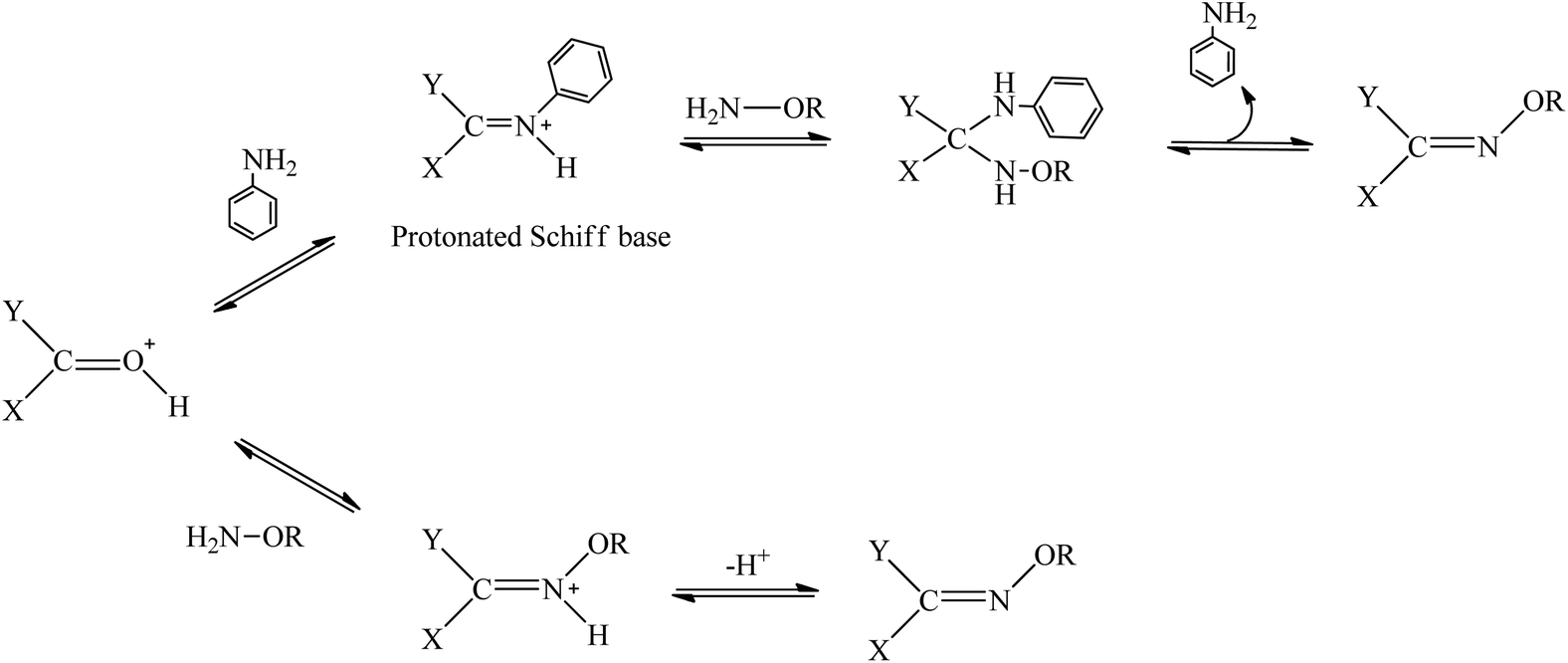
The proposed mechanism of salt-catalysed oxime formation. (a) Reaction... | Download Scientific Diagram
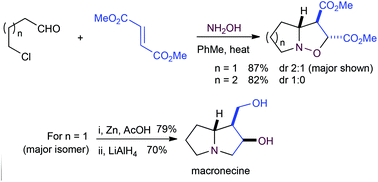
Cascade oxime formation, cyclization to a nitrone, and intermolecular dipolar cycloaddition - Organic & Biomolecular Chemistry (RSC Publishing)
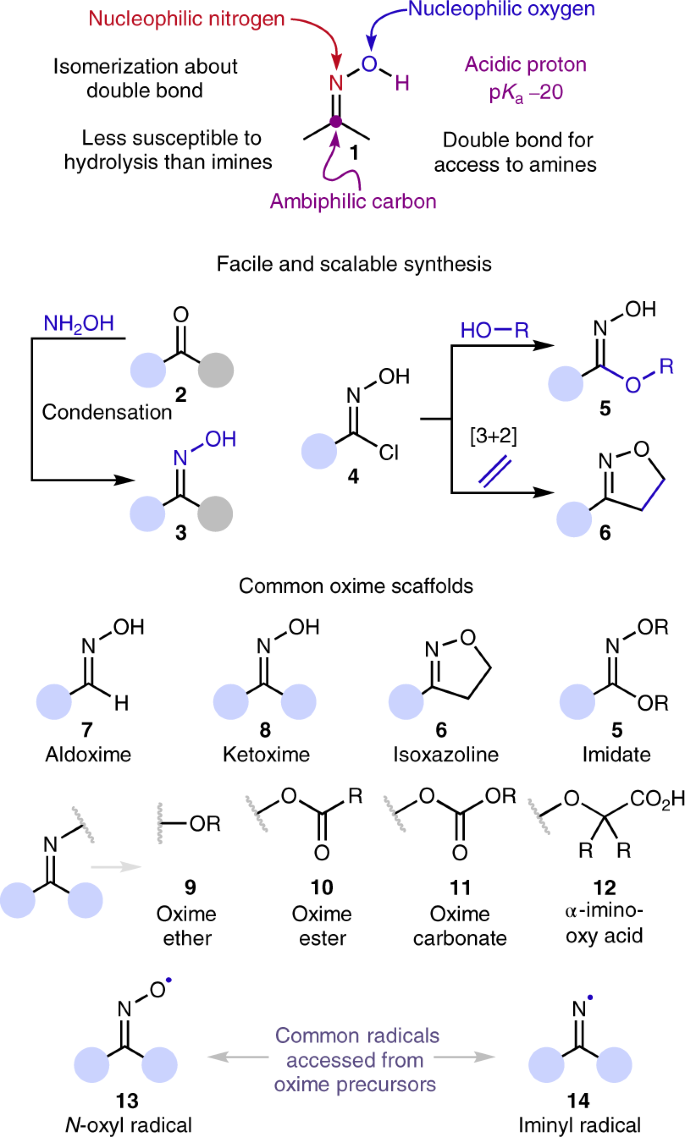
The emergence of oxime click chemistry and its utility in polymer science - Polymer Chemistry (RSC Publishing) DOI:10.1039/C6PY00635C