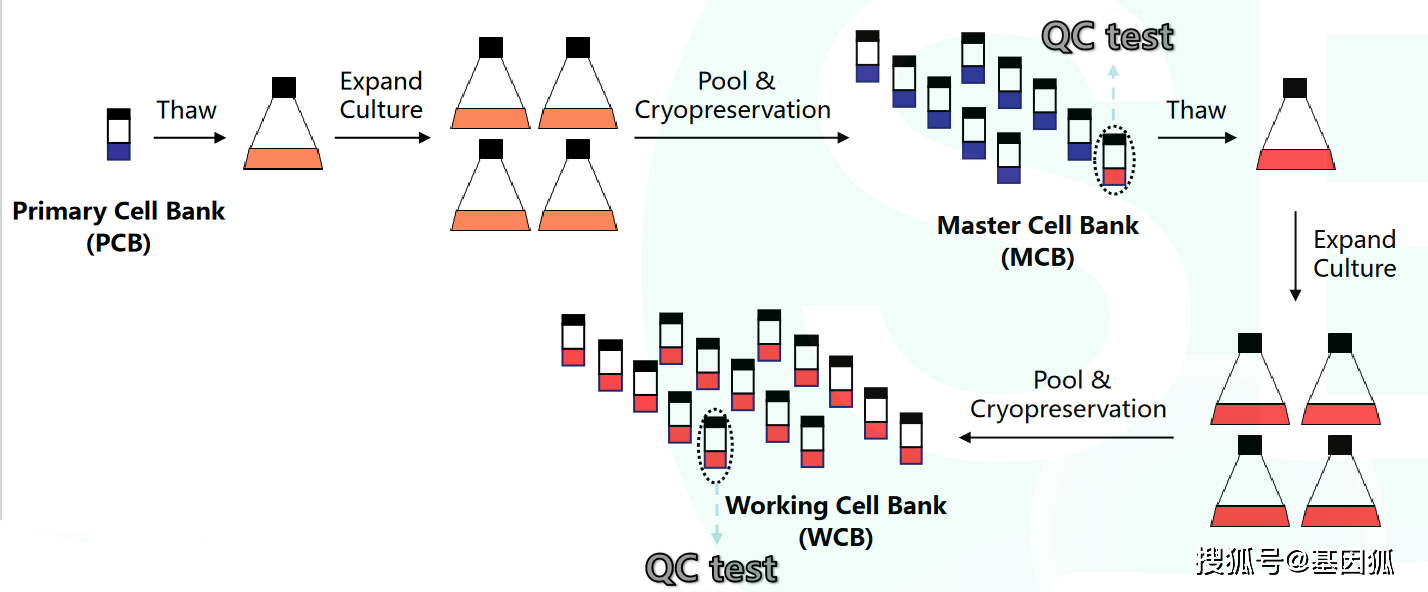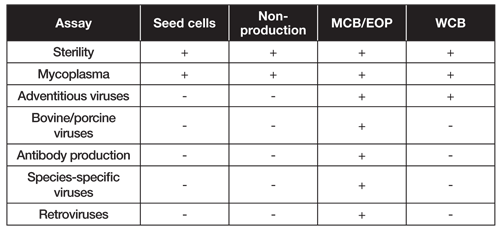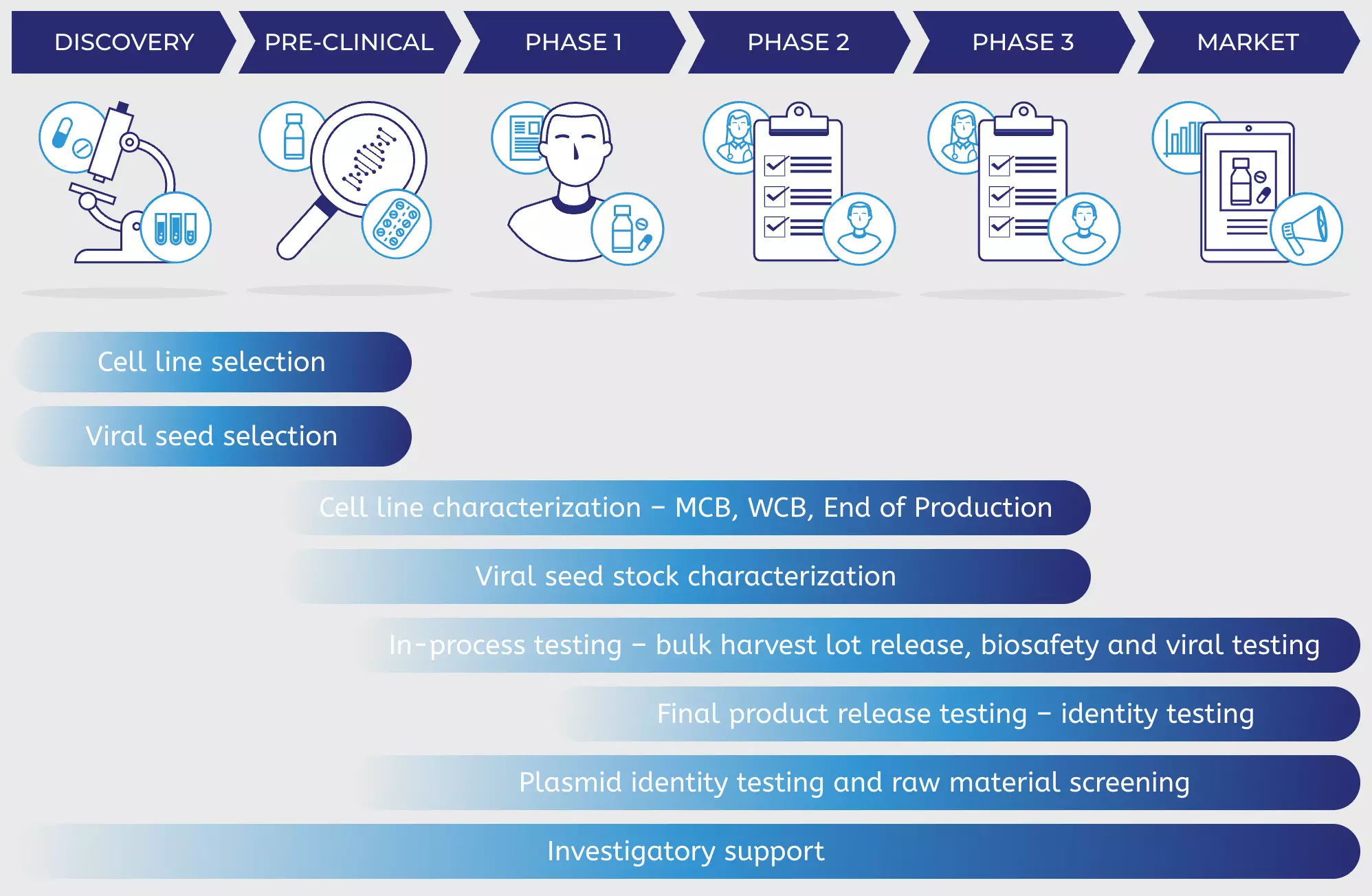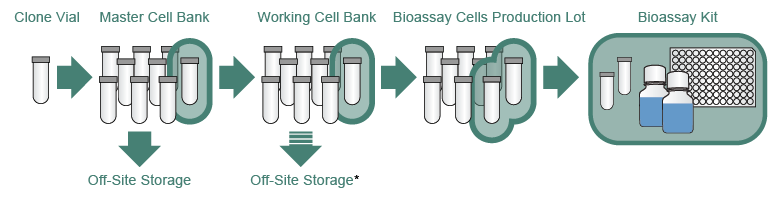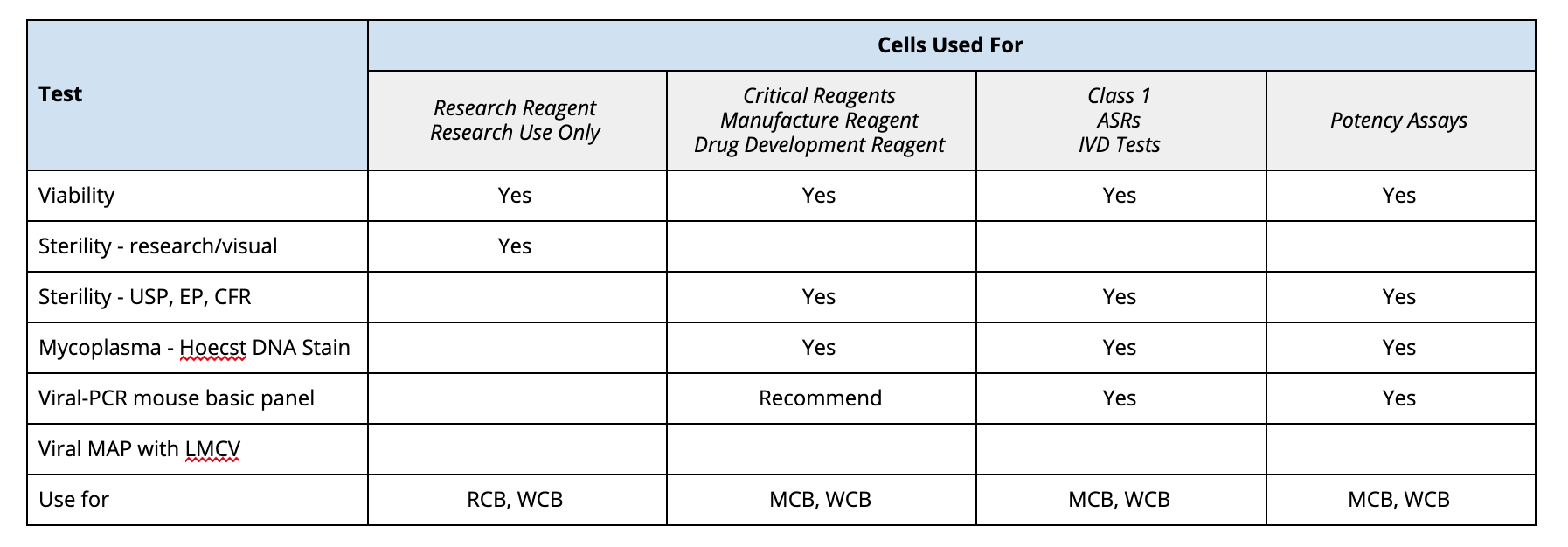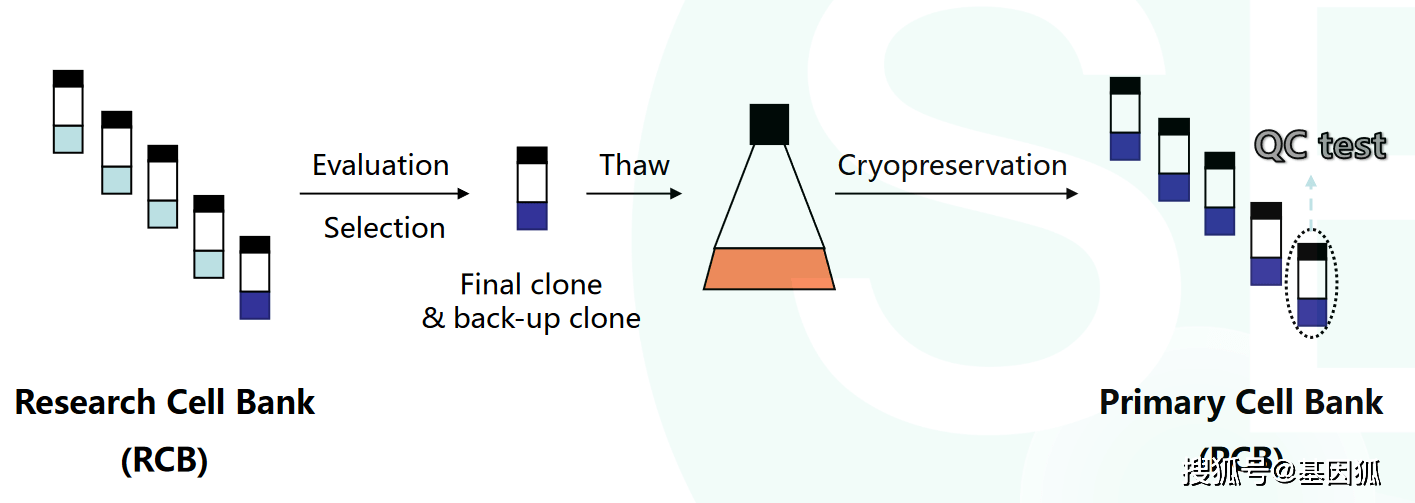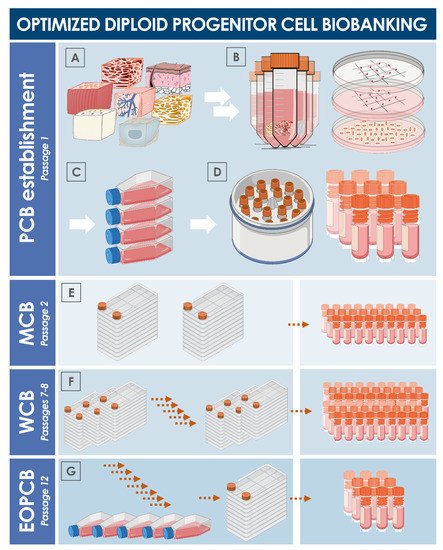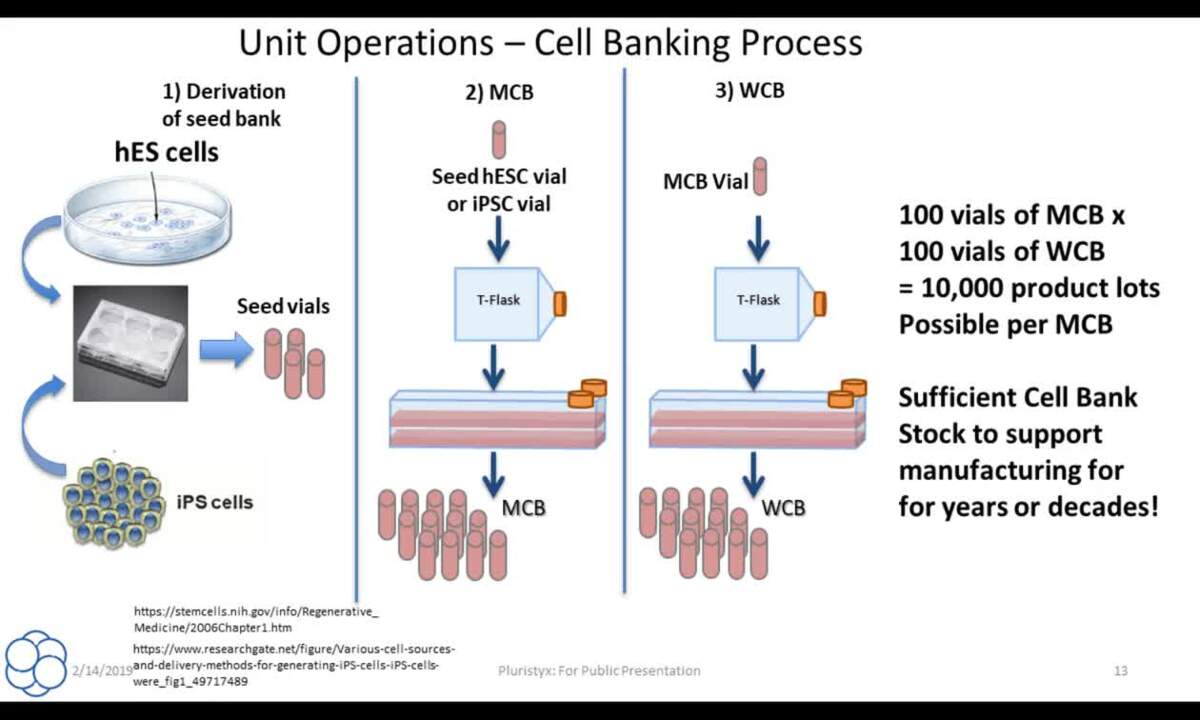
GMP cell banks as part of a staged, standardized, platform-style cell production process - Scientific Videos | Thermo Fisher Scientific US

GMP Tiered Cell Banking of Non-enzymatically Isolated Dermal Progenitor Fibroblasts for Allogenic Regenerative Medicine | SpringerLink

Cell Banking of HEK293T cell line for clinical-grade lentiviral particles manufacturing | Translational Medicine Communications | Full Text

A Roadmap for the Production of a GMP-Compatible Cell Bank of Allogeneic Bone Marrow-Derived Clonal Mesenchymal Stromal Cells for Cell Therapy Applications | Stem Cell Reviews and Reports
Consistency and Safety of Cell Banks for Research and Clinical Use: Preliminary Analysis of Fetal Skin Banks
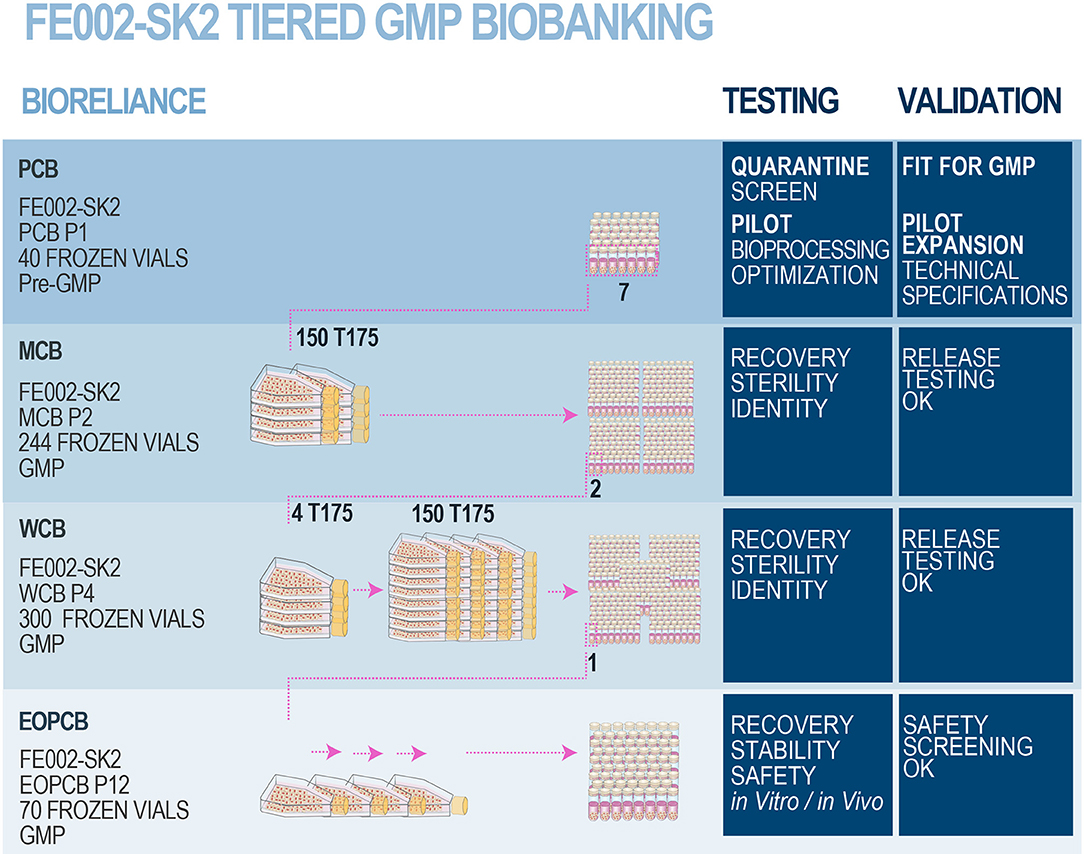
Frontiers | Bringing Safe and Standardized Cell Therapies to Industrialized Processing for Burns and Wounds
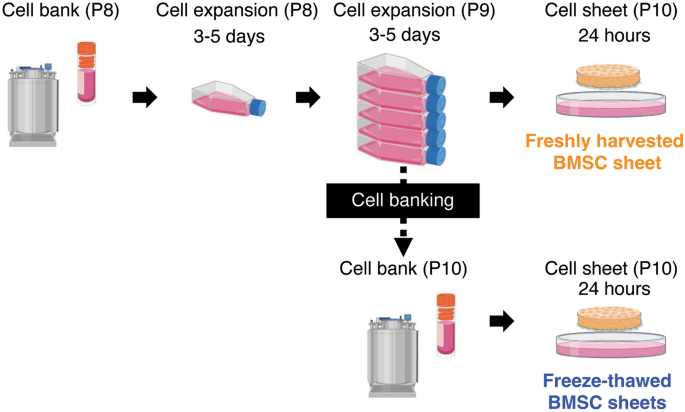
Rapid and effective preparation of clonal bone marrow-derived mesenchymal stem/stromal cell sheets to reduce renal fibrosis | Scientific Reports