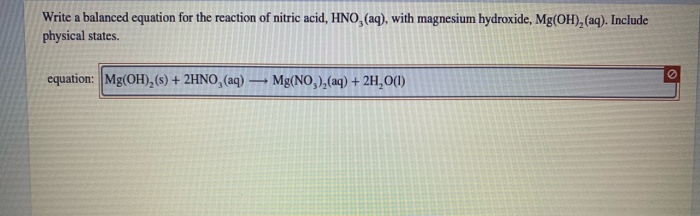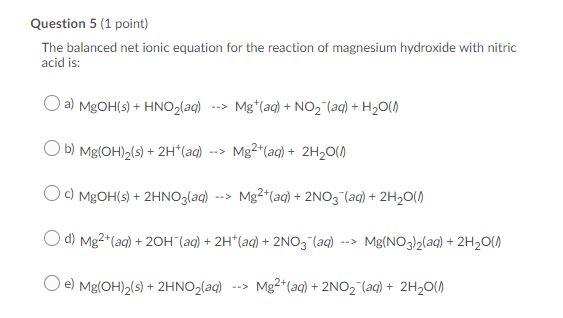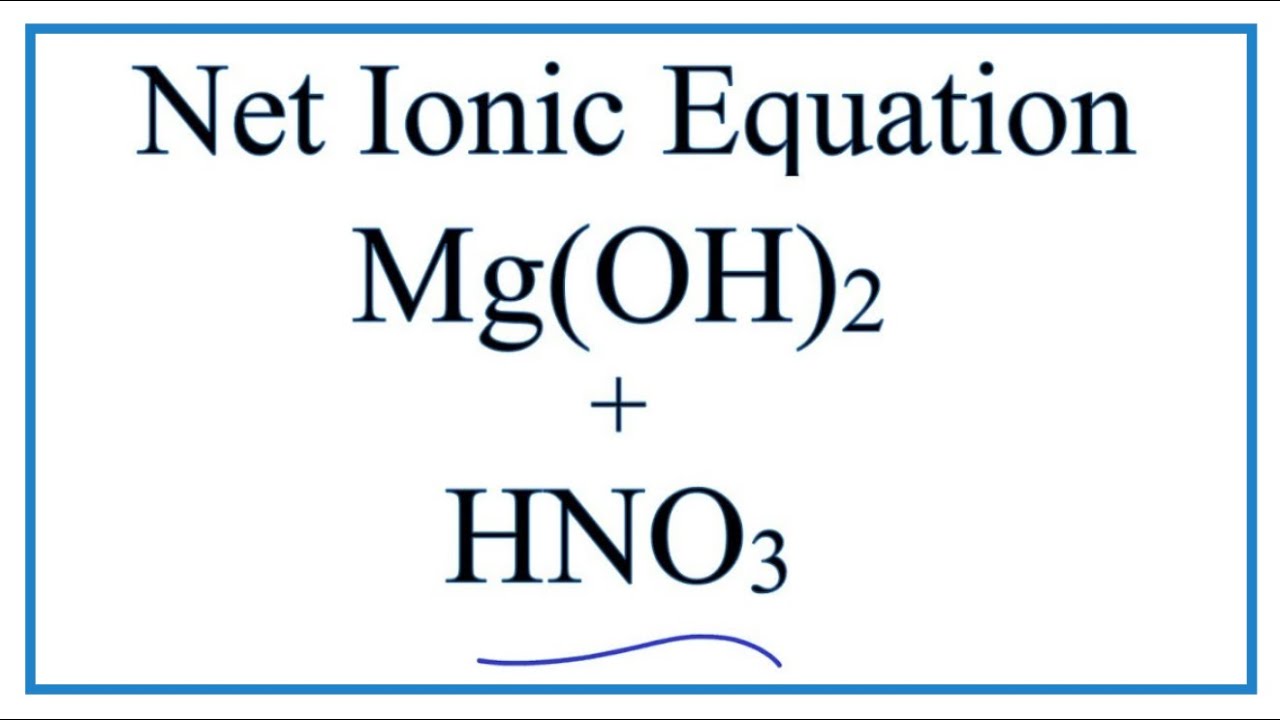Match up the substance with the pH Lemon juice Hydrochloric acid Soap Sodium hydroxide Water pH 14 pH 0 pH 3 pH 7 pH ppt download

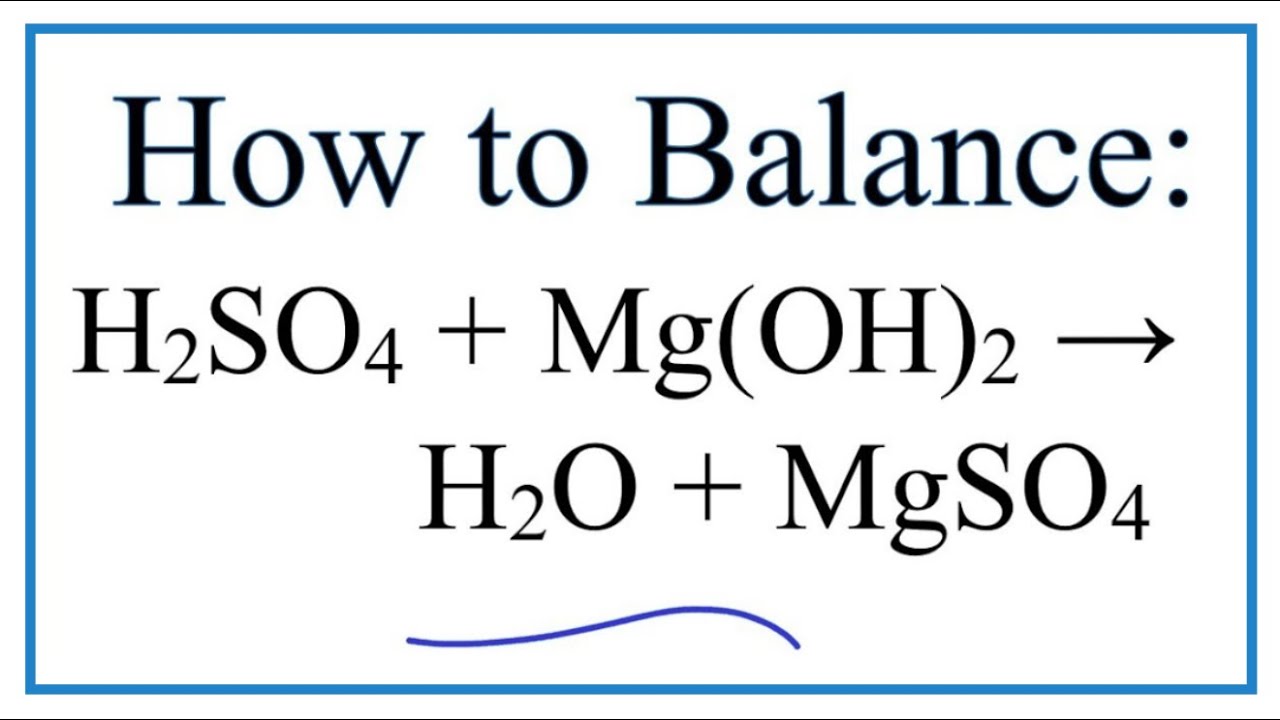
Mg(OH)2+HNO3=Mg(NO3)2+H2O Balanced Equation||Magnesium Hydroxide+Nitric acid =Magnesium Nitrate+Water - YouTube

SOLVED: Magnesium nitrate can be produced from the reaction of magnesium hydroxide and nitric acid according to the reaction: MBIOHIz 2 HNOg MB(NOzh 2 Hzo How many grams of MB(NOah can be

Solution For: Write two equations for the neutralization of nitric acid, HNO, with magnesium hydroxide, - Brainly.com