Most Online Prospecting Physics | Chemistry Download List of Practicals & Experiment 2020 of CBSE RBSE NCERT Examinations
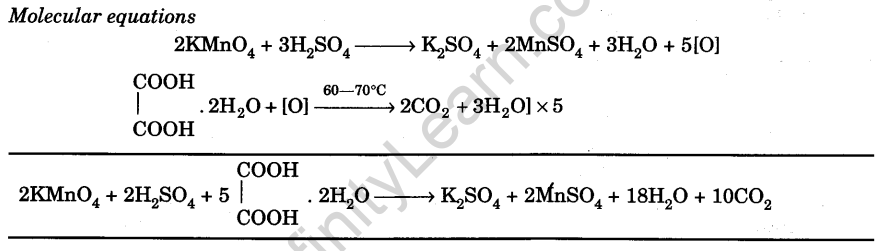
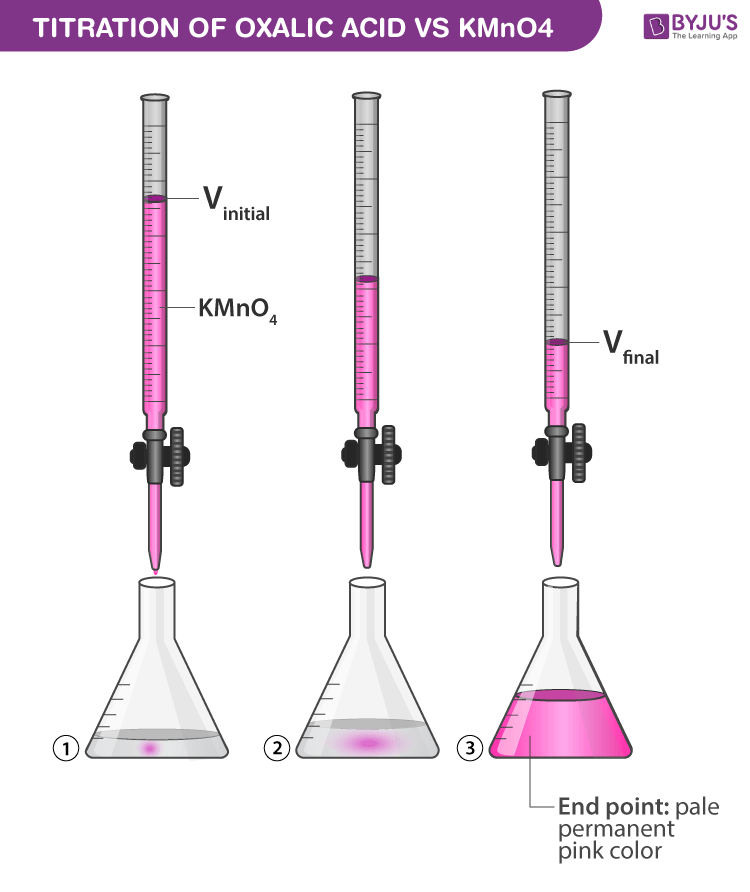
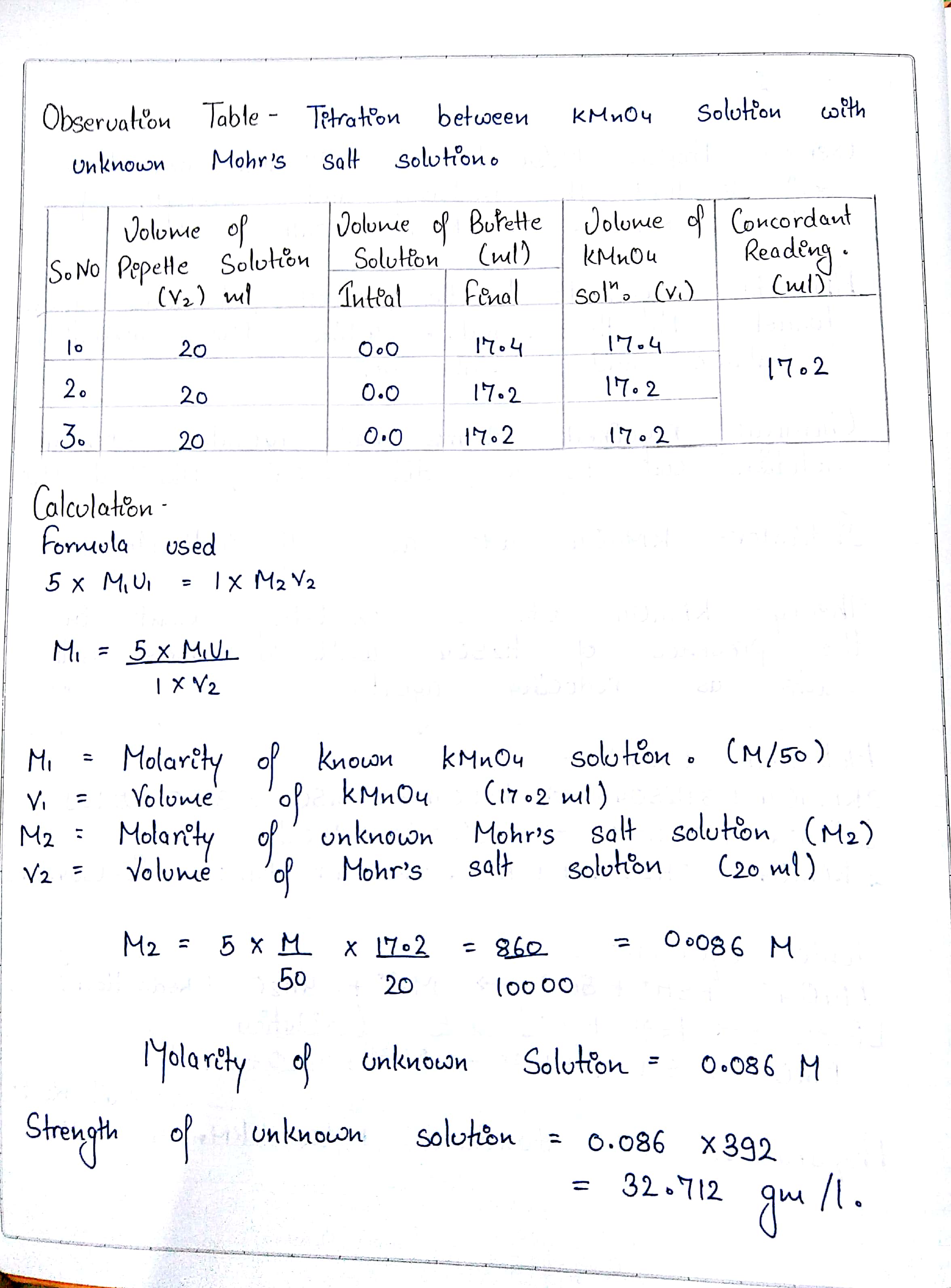
Prepare M/50 Solution of Oxalic Acid. With its help, Determine 50 the Molarity and Strength of the Given Solution of Potassium Permanganate - Infinity Learn

Potassium permanganate in presence of dilute H2SO4 reacts with oxalic acid (anhydrous). Write the balanced equation of the reaction and find out how many moles of oxalic acid are oxidised by 2

SOLUTION: Experiment prepare m 50 solution of oxalic acid with its help determine 50 the molarity and strength of the given solution of potassium permanganate kmno4 - Studypool

SOLVED: A solution of potassium permanganate is standardized by titration with oxalic acid (HzC204) It required 28.97mL of the permanganate solution to completely react with 0.1058g of oxalic acid (MW = 90.03

Titration- Oxalic Acid Vs KMnO4 in Hindi | Full Experiment with Calculations - Chemistry Practical - YouTube
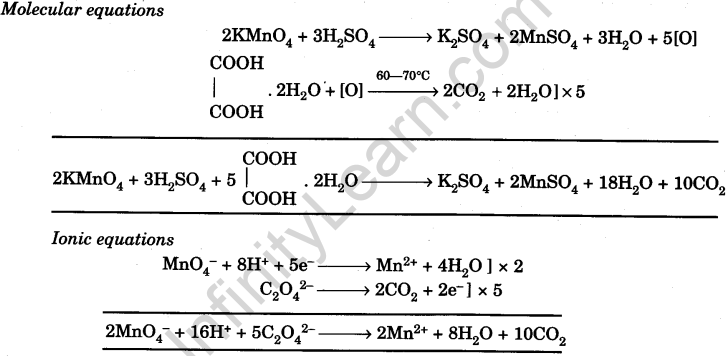
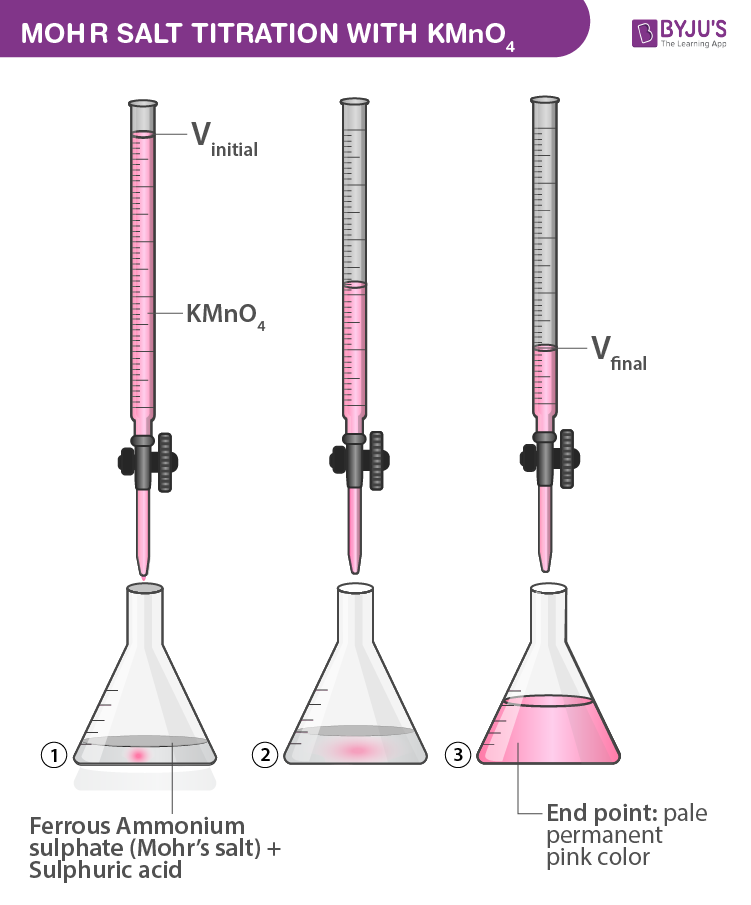
Prepare N/20 solution of oxalic acid. Using this solution, find out strength and normality of the given potassium permanganate solution

In the titration between oxalic acid and acidified potassium permanganate ,the manganous salt formed during the reaction and catalysis the reaction .The manganous salts acts as :
Amount of oxalic acid present in a solution can be determined by its titration with KMnO4 solution in the presence of H2SO4. - Sarthaks eConnect | Largest Online Education Community