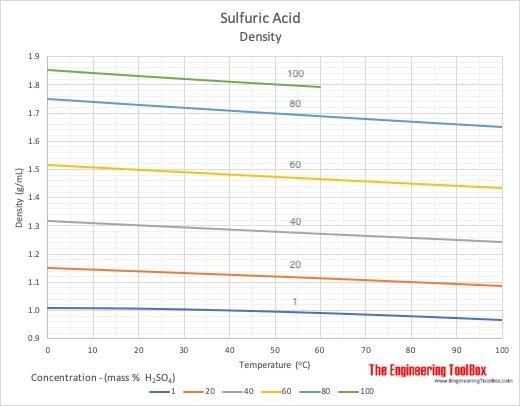
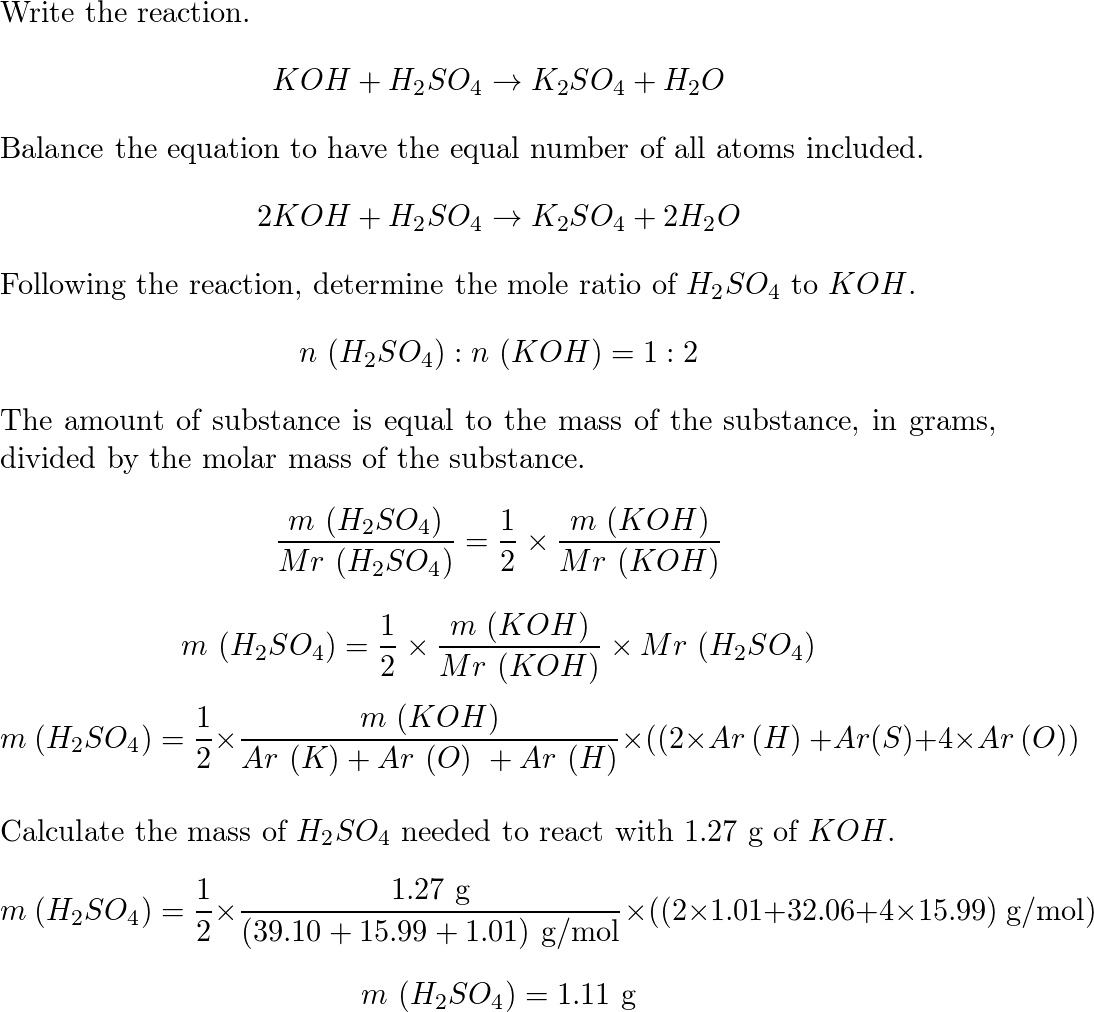Corrosion properties of mild steel in 1M H2SO4 solution at 313 K in the... | Download Scientific Diagram

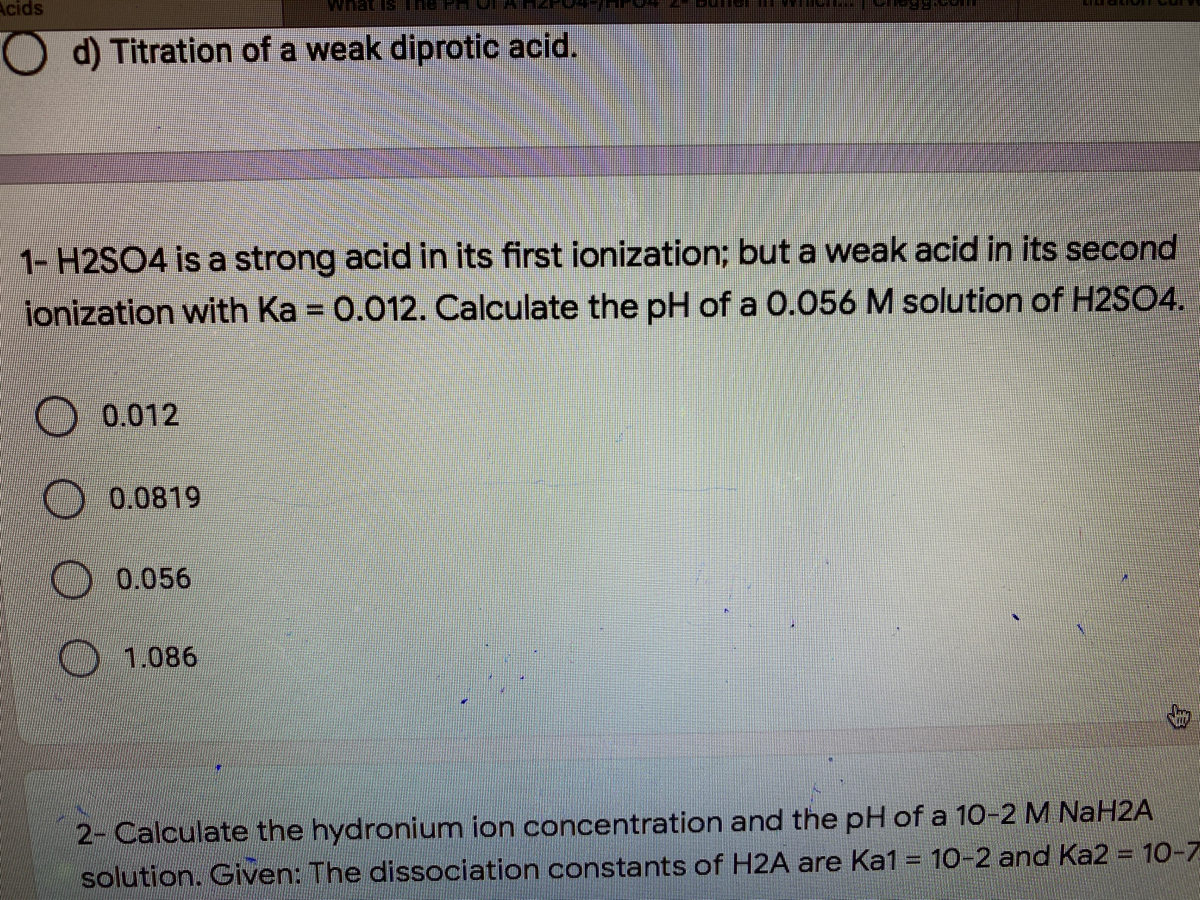
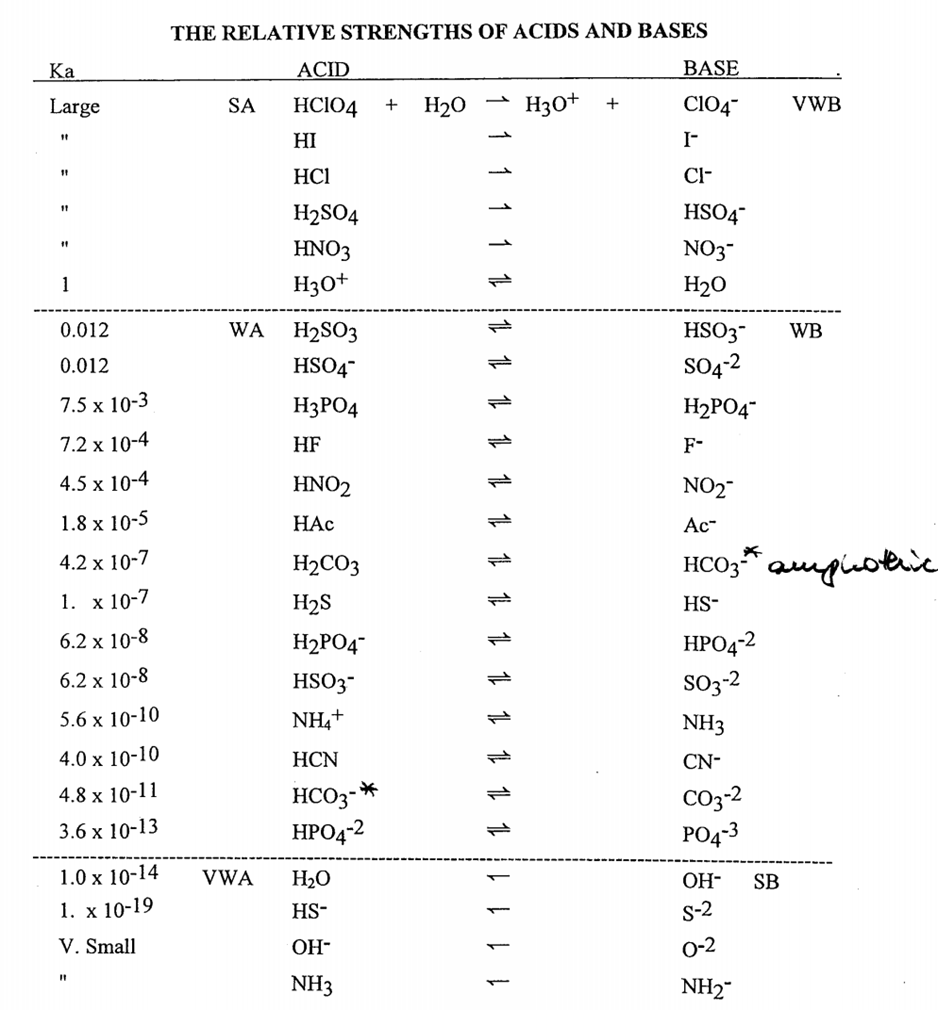
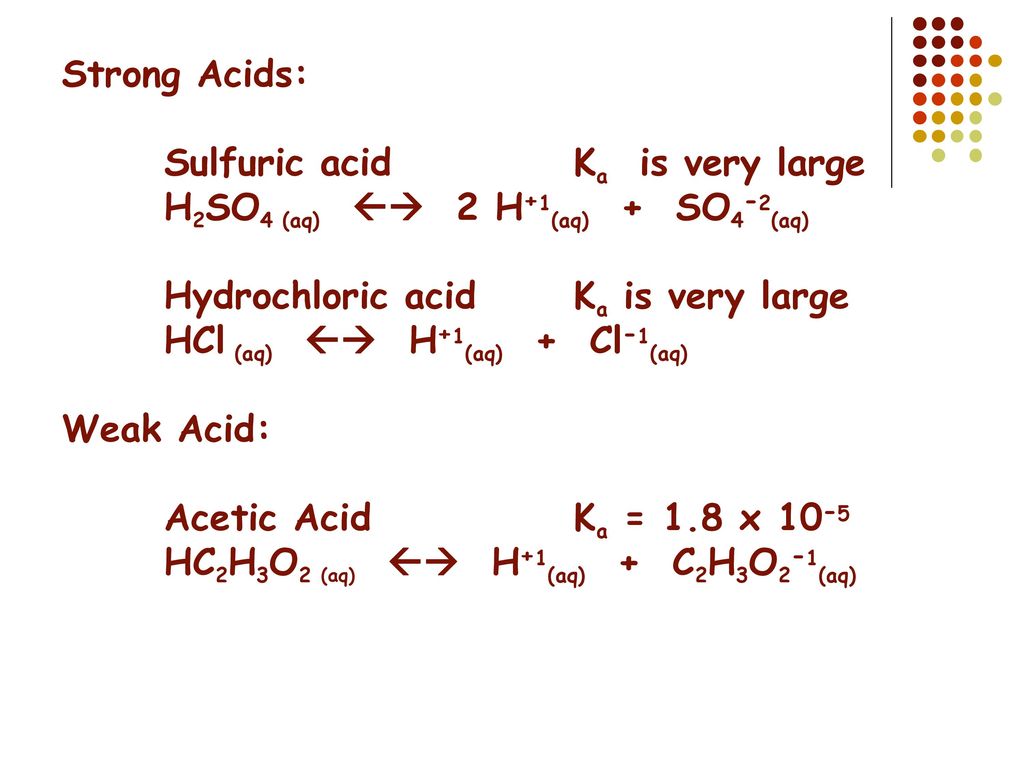
If Ka1 and Ka2 of sulphuric acid are 1 × 10^-2 and 1 × 10^-6 respectively, then concentration of sulphate ions in 0.01 MH2SO4 solution will be:

Question 26 of 32What is the equation for the acid dissociation constant, Ka, of carbonic acid? - brainly.com