Practical guidance for procedures related to Brexit for medicinal products for human use approved via MRP/DCP

Part III - Data Interpretation and Application | Supporting Materials for NCHRP Report 626 |The National Academies Press

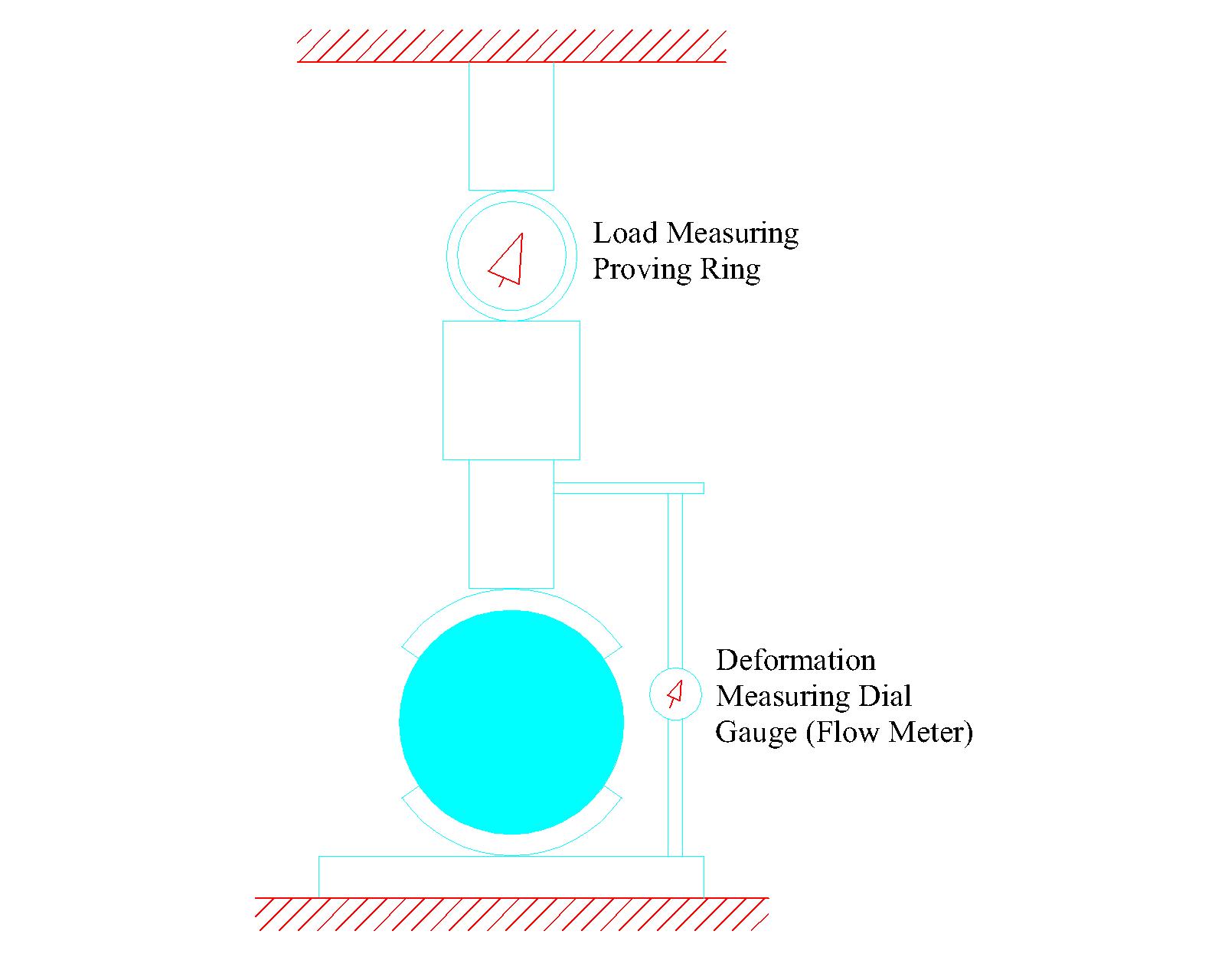
Appendix A - Glossary | NDT Technology for Quality Assurance of HMA Pavement Construction |The National Academies Press
We are happy to welcome you, also in the name of the STARS consortium, to the first free online Curriculum teaching regulatory s

Chapter 2 - Materials Testing for Construction Quality Determination | NDT Technology for Quality Assurance of HMA Pavement Construction |The National Academies Press
Norwegian guideline for packaging of human and veterinary medicinal products with marketing authorisation/registration
European Medicines Agency pre-authorisation procedural advice for users of the centralised procedure
Q&A - List for the submission of variations for human medicinal products according to Commission Regulation (EC) 1234/2008
Appendix A - Glossary | NDT Technology for Quality Assurance of HMA Pavement Construction |The National Academies Press