Given that the relative molecular mass of copper oxide is 80 , what volume of ammonia measured at STP is required to completely reduce 120 g of copper oxide? The equation for

When ammonia is passed over heated copper oxide, the metallic coper is obtained. The reaction shows - YouTube

Copper based catalysts for the selective ammonia oxidation into nitrogen and water vapour—Recent trends and open challenges - ScienceDirect

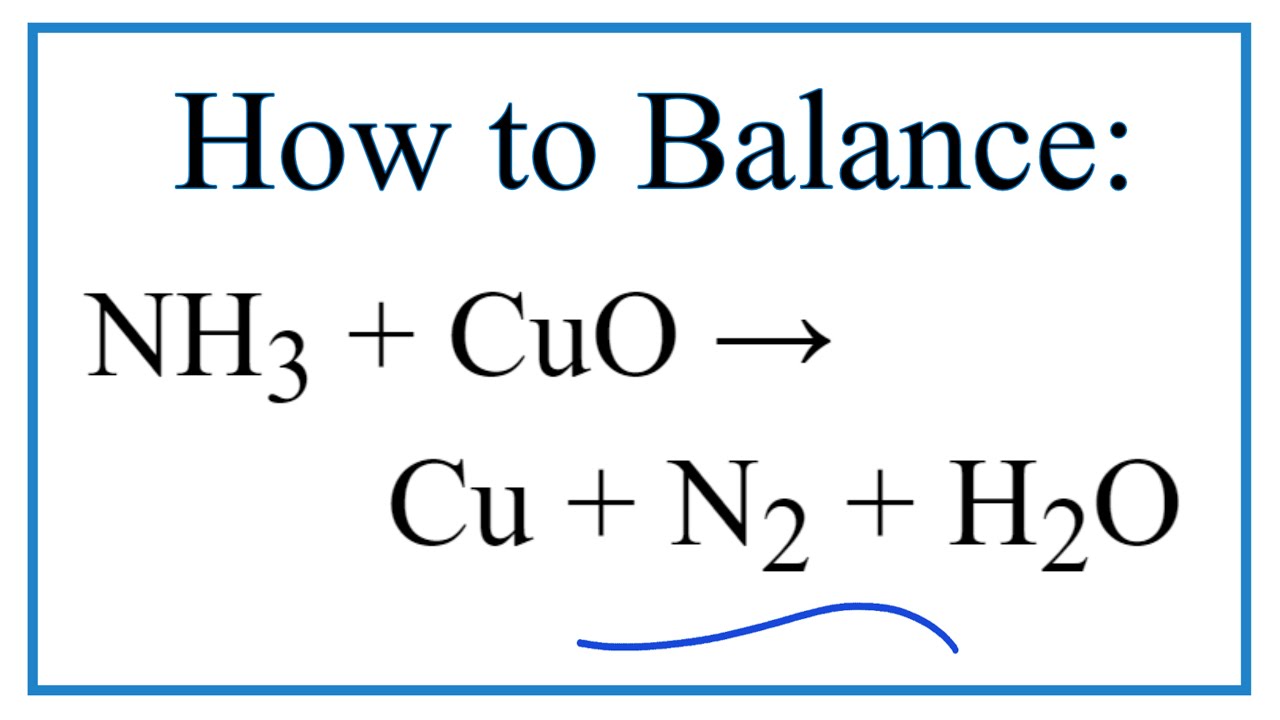
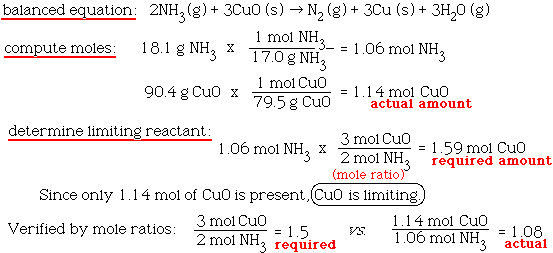
Copper(II) oxide reacts with ammonia to give copper, water and nitrogen. CuO((s)) +NH(3(g)) to xCu((s))+yH(2)O((l)) +zN(2(g)) The value of (x + y + z) is

When ammonia is passed over heated copper oxide, the metallic coper is obtained. The reaction shows - YouTube

When ammonia is passed over heated copper oxide, the metallic copper is obtained, the reaction shows - YouTube

Give balanced equation for each of the following:Reduction of hot Copper (II) oxide to copper using ammonia gas:
![State one obervation for the following : Ammonia gas is passed over heated copper [II] oxide. | ... - YouTube State one obervation for the following : Ammonia gas is passed over heated copper [II] oxide. | ... - YouTube](https://i.ytimg.com/vi/7deWf3VEtQk/maxresdefault.jpg)
State one obervation for the following : Ammonia gas is passed over heated copper [II] oxide. | ... - YouTube
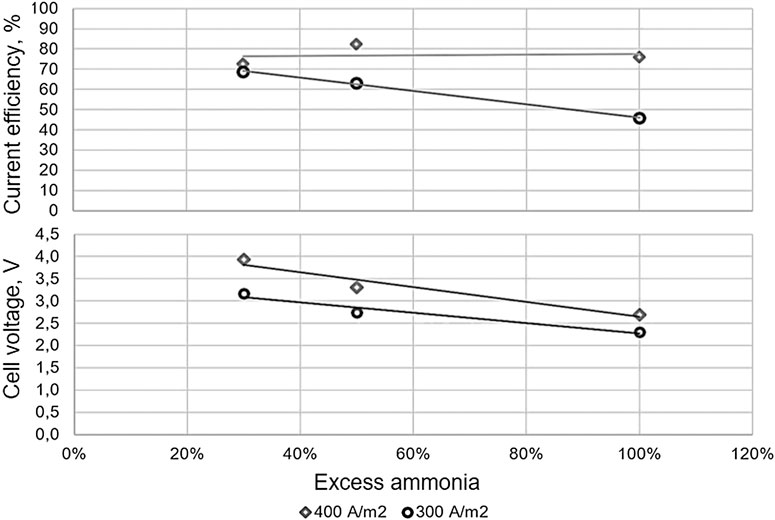
![PDF] Dissolution kinetics of low grade complex copper ore in ammonia- ammonium chloride solution | Semantic Scholar PDF] Dissolution kinetics of low grade complex copper ore in ammonia- ammonium chloride solution | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/74104461ce1759e6c90926cb2fd02cdd1b4c9a35/2-Table1-1.png)
PDF] Dissolution kinetics of low grade complex copper ore in ammonia- ammonium chloride solution | Semantic Scholar

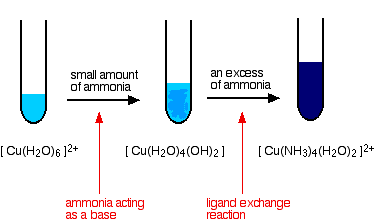
Electrocatalytic, Homogeneous Ammonia Oxidation in Water to Nitrate and Nitrite with a Copper Complex | Journal of the American Chemical Society

Selective catalytic oxidation of ammonia to nitric oxide via chemical looping | Nature Communications
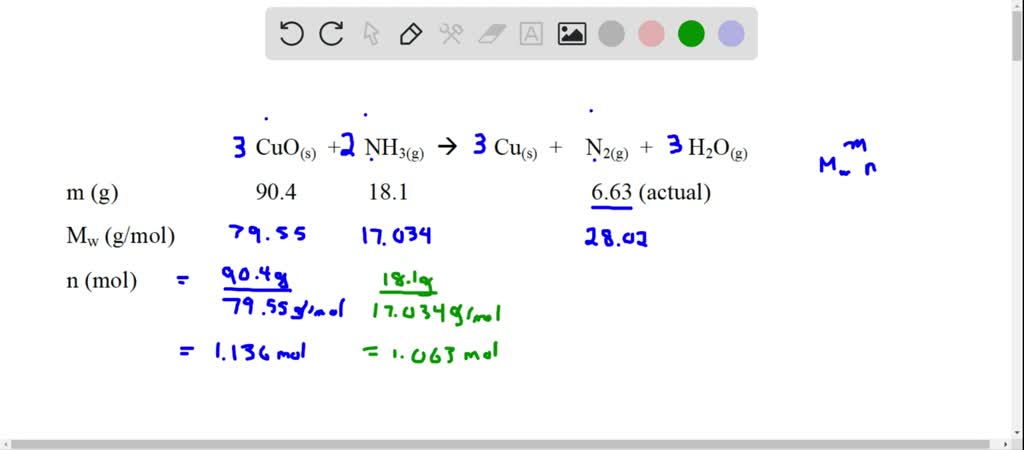
SOLVED: Gaseous ammonia, NH3, and copper(II) oxide, CuO, react together to form gaseous nitrogen (N2), solid copper, and water vapor. If a sample containing 17.9 g of NH3is reacted with 90.0 g