Table 1 from Conversion of Natural Aldehydes from Eucalyptus citriodora, Cymbopogon citratus, and Lippia multiflora into Oximes: GC-MS and FT-IR Analysis † | Semantic Scholar

Natural Product The organic compounds isolated from the living organism i.e plants, animals and micro organism are generally known as Natural products. - ppt download
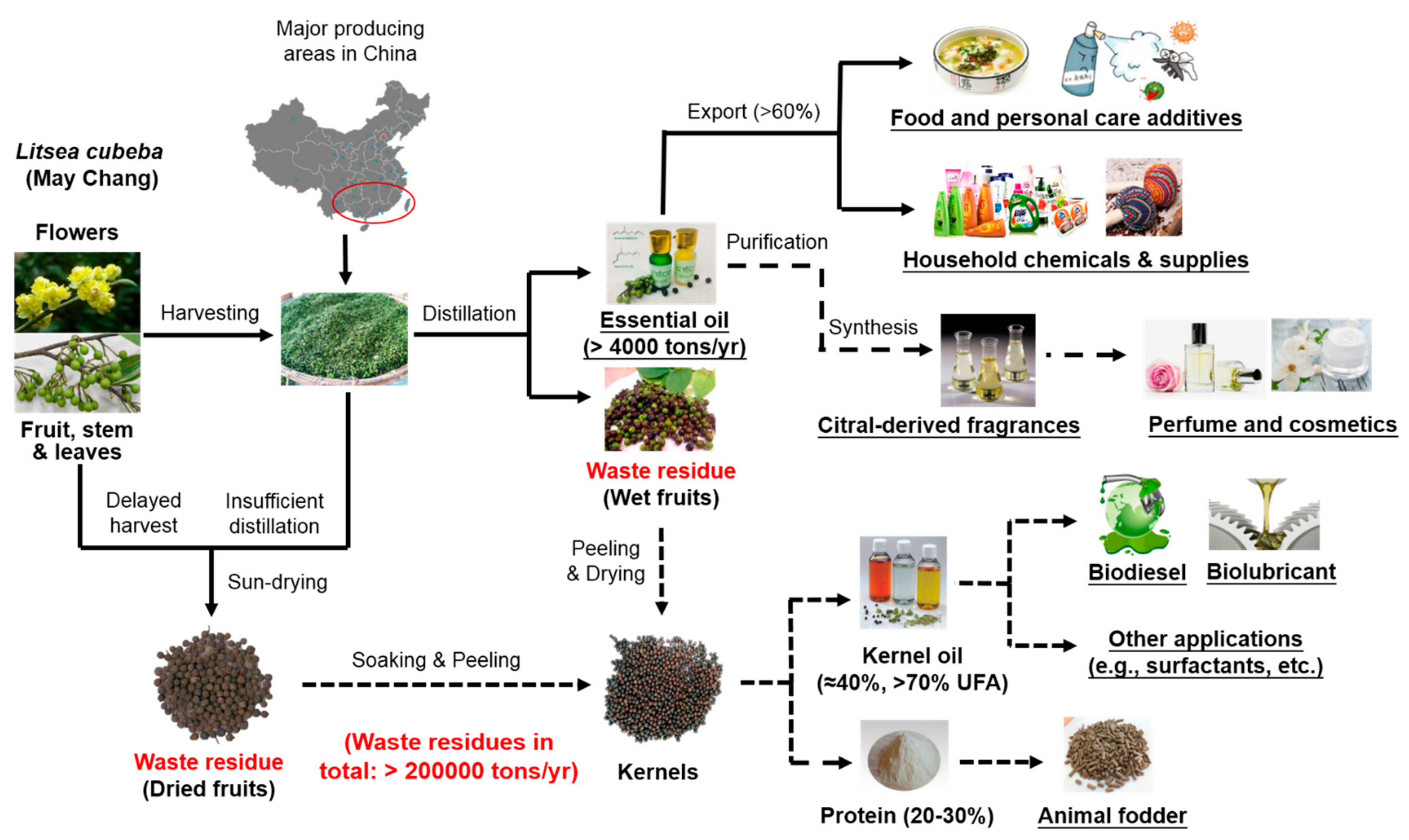
Molecules | Free Full-Text | An Overview on Total Valorization of Litsea cubeba as a New Woody Oil Plant Resource toward a Zero-Waste Biorefinery

Oximes of 3,7-dimethylocta-2,6-dienal: Green synthesis, preparative separation of all diastereomers and complete assignment of 1H and 13C NMR spectra - ScienceDirect

Oximes of 3,7-dimethylocta-2,6-dienal: Green synthesis, preparative separation of all diastereomers and complete assignment of 1H and 13C NMR spectra - ScienceDirect

Toward the Efficient Synthesis of Pseudoionone from Citral in a Continuous-Flow Microreactor | Industrial & Engineering Chemistry Research