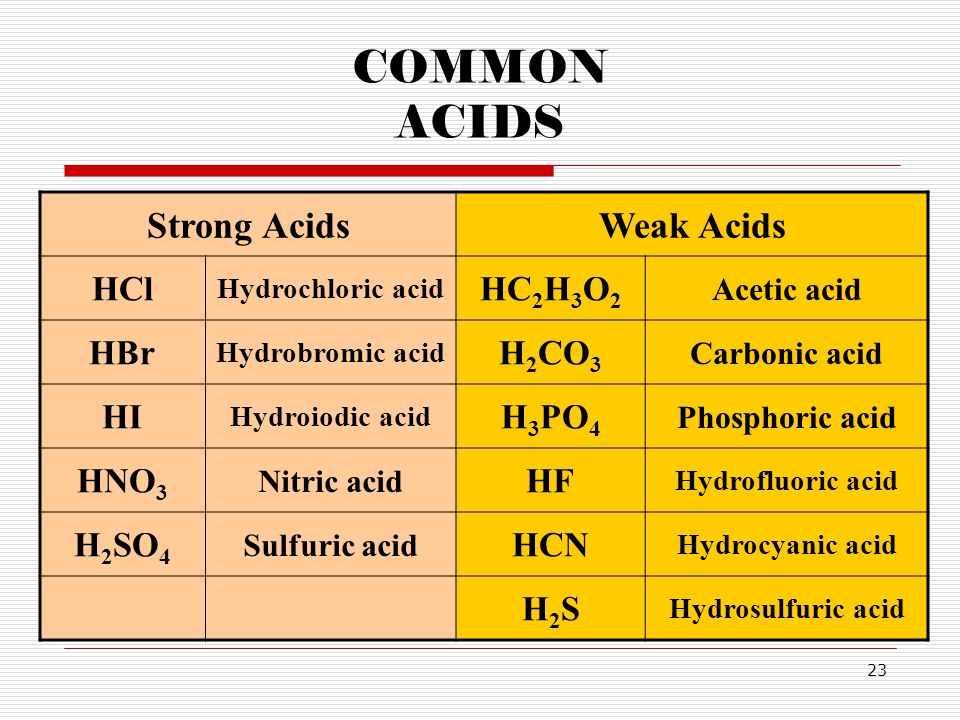
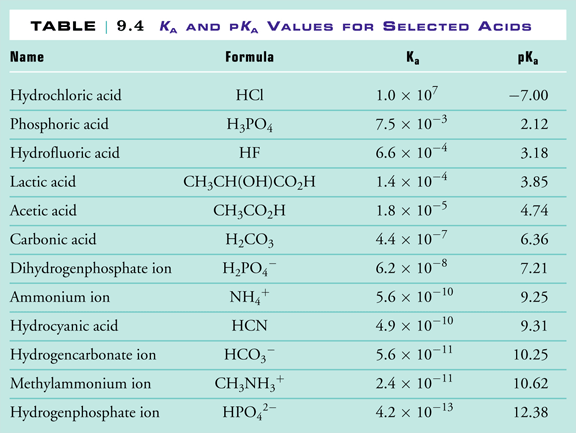
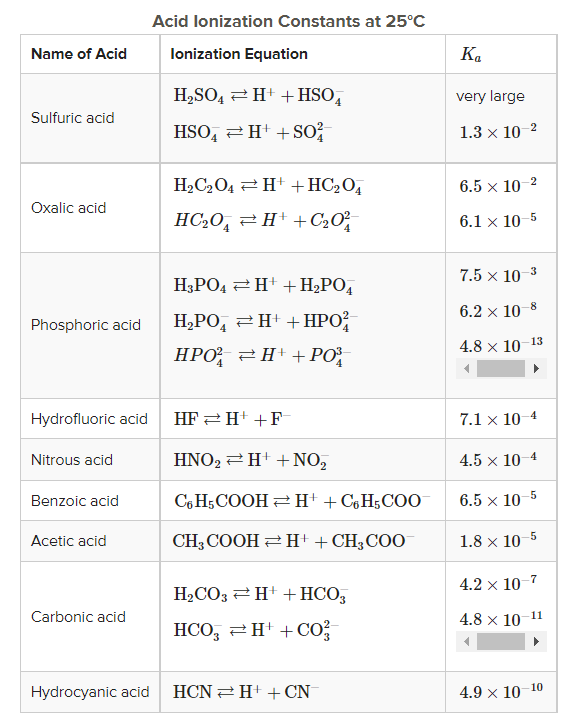
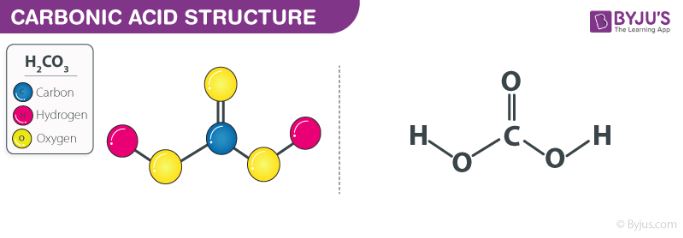
Why is carbonic acid a weak acid even though it gets completely dissociated into H+ and CO3- ions? - Quora
Why is carbonic acid a weak acid even though it gets completely dissociated into H+ and CO3- ions? - Quora

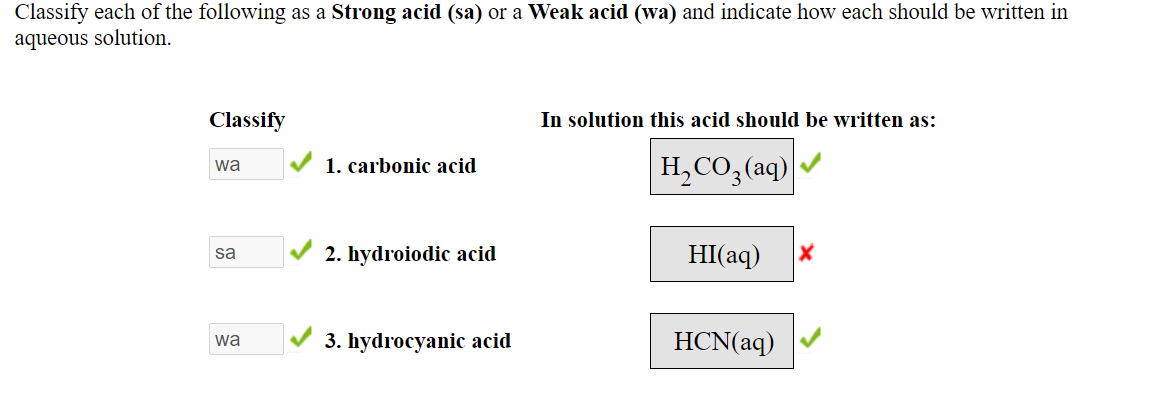
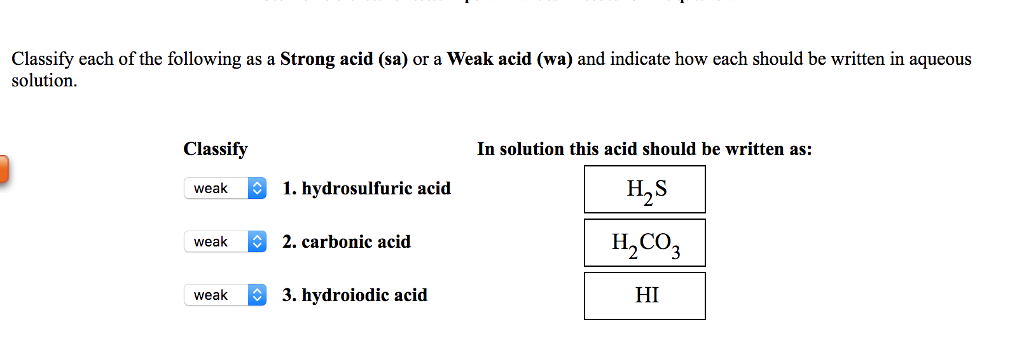
SOLVED: Classify each of the following as Strong acid (sa) or Weak acid (wa) and indicate how each should be written in aqueous solution. Classify In solution this acid should be written
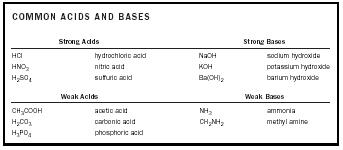
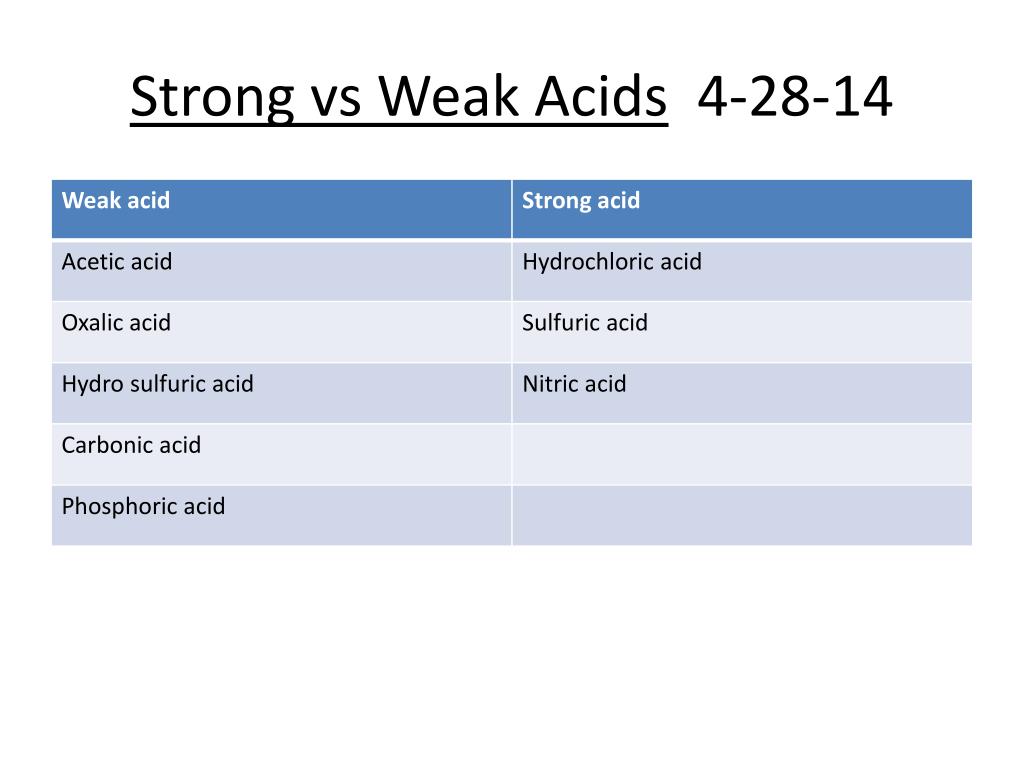
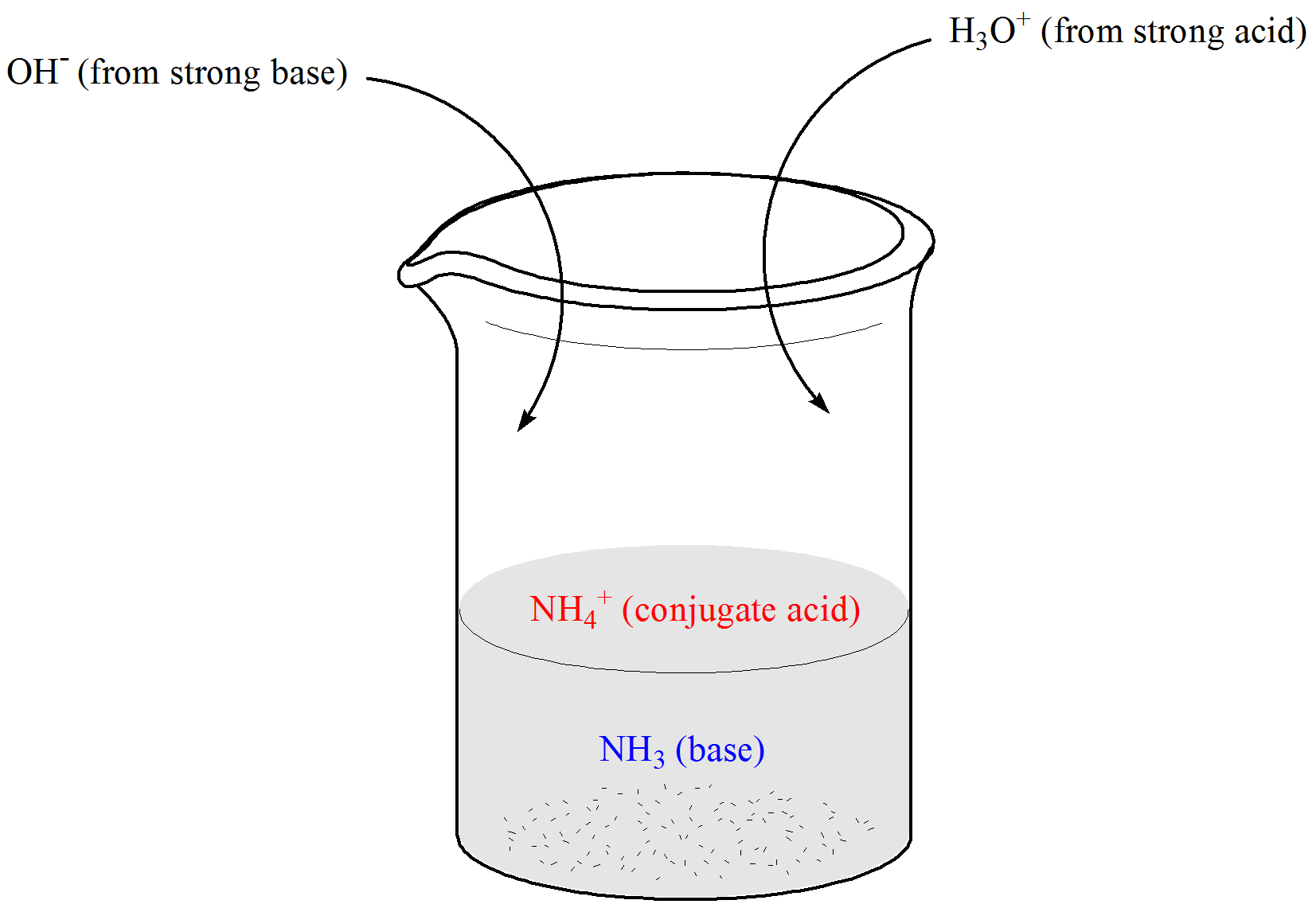
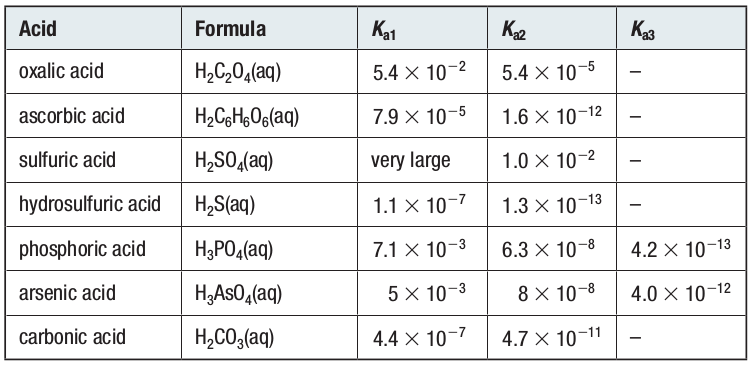
Acid-Base Chemistry - Chemistry Encyclopedia - reaction, water, metal, gas, number, equation, salt, property



:max_bytes(150000):strip_icc()/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)