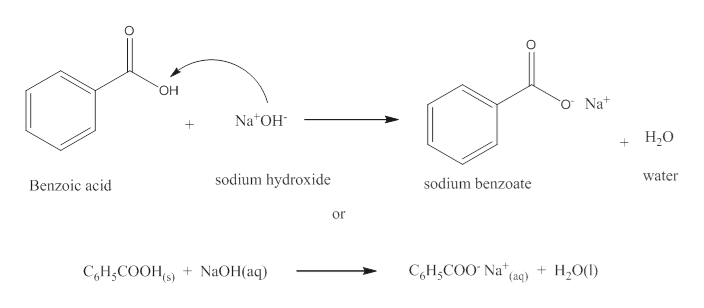
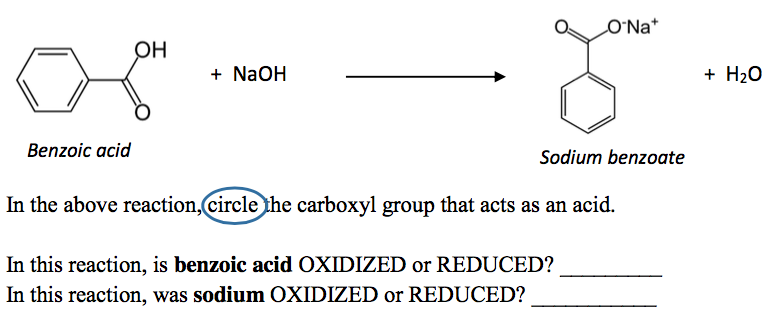✓ Solved: When benzoic acid ( 5 ) is partitioned between diethyl ether and aqueous sodium hydroxide solution...

Explain the results for the tube in which 1.0 m naoh was added to benzoic acid. write an equation for this, - Brainly.com

Draw the products of benzoic acid reacting with sodium hydroxide. Draw the products of the pyridine reacting with hydrochloric acid. Use the "+/-" button to add the charge (and H atom).

Write a balanced equation for the reaction of benzoic acid with hydroxide ion. Why is it necessary to extract the ether layer with sodium hydroxide? | Homework.Study.com
![Which of the following will give phenol with \\[{\\text{CaO}}\\] and \\[{\\text{NaOH}}\\]?A. Salicylic acidB. Picric acidC. Benzoic acidD. Amino acid Which of the following will give phenol with \\[{\\text{CaO}}\\] and \\[{\\text{NaOH}}\\]?A. Salicylic acidB. Picric acidC. Benzoic acidD. Amino acid](https://www.vedantu.com/question-sets/39d15196-f3b7-4d8c-bde9-5684a8a019909186147776397460082.png)
Which of the following will give phenol with \\[{\\text{CaO}}\\] and \\[{\\text{NaOH}}\\]?A. Salicylic acidB. Picric acidC. Benzoic acidD. Amino acid

State, whether the following statements are True or False:Benzamide on treatment with NaOH and Br2 forms benzoic acid.
✓ Solved: Explain the results for the tube in which 1.0 M NaOH was added to benzoic acid. Write an equation...

When a solution of benzoic acid was titrated with `NaOH` the `pH` of the solution when half - YouTube

Write the mechanism for the reaction of either benzoic acid or acetic acid with NaOH. Be sure to include all major structures and resonance forms. | Homework.Study.com