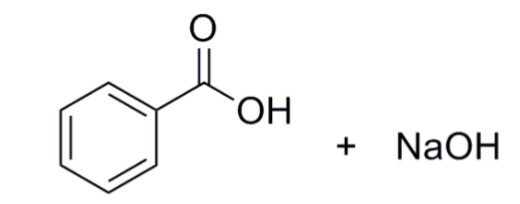
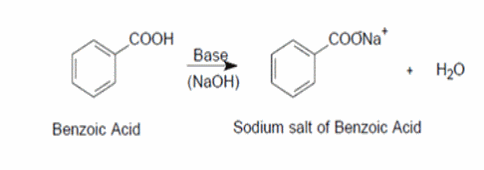
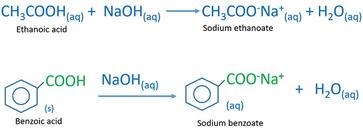
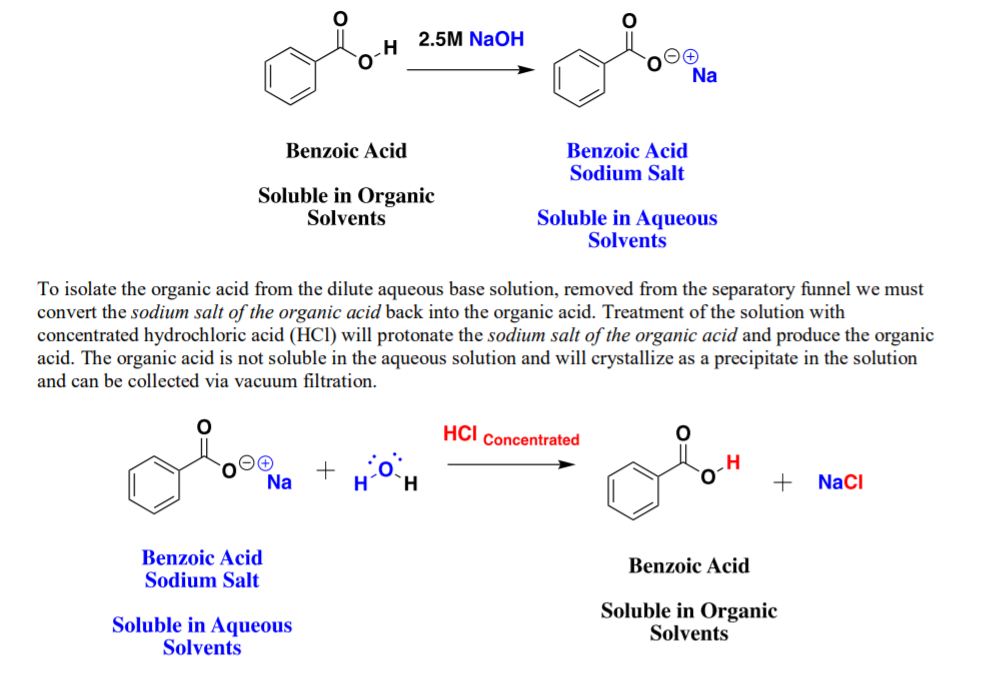
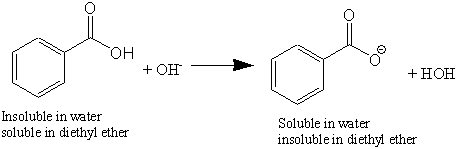
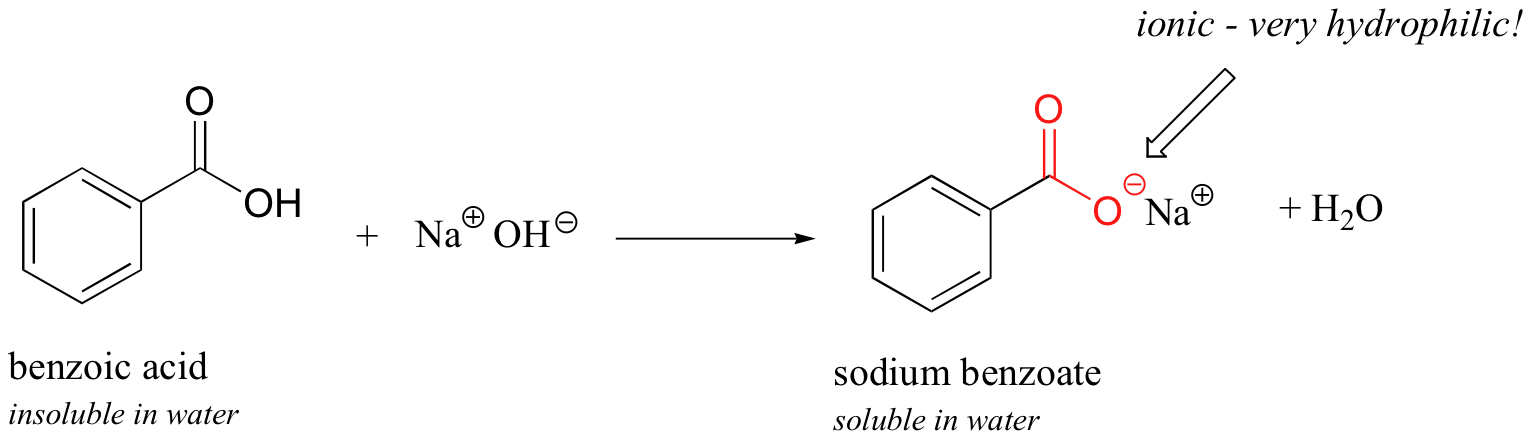
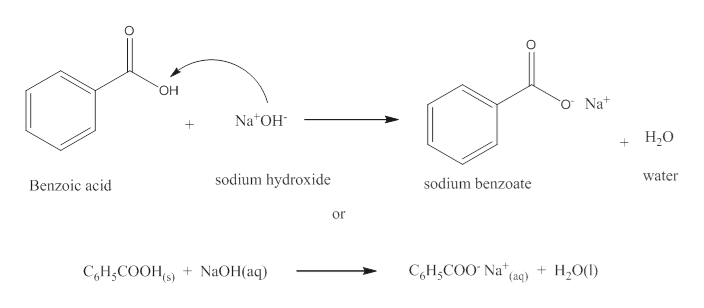
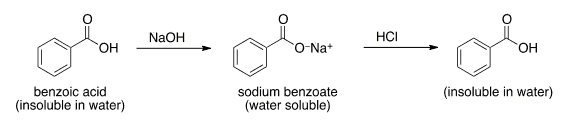
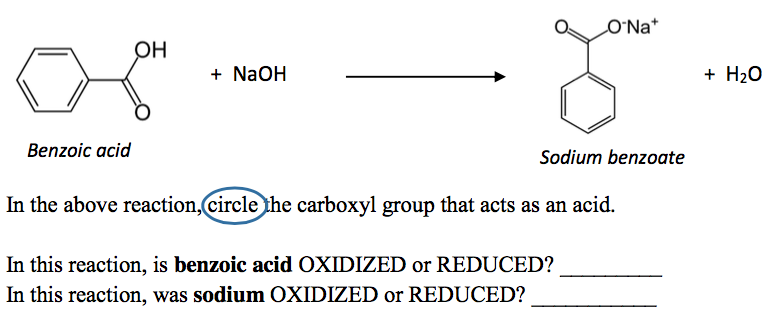1. Draw a balanced chemical equation for the reaction that would occur between benzoic acid and aqueous sodium hydroxide. 2. Draw a balanced chemical equation for the reaction that would occur between

When a solution of benzoic acid was titrated with `NaOH` the `pH` of the solution when half - YouTube
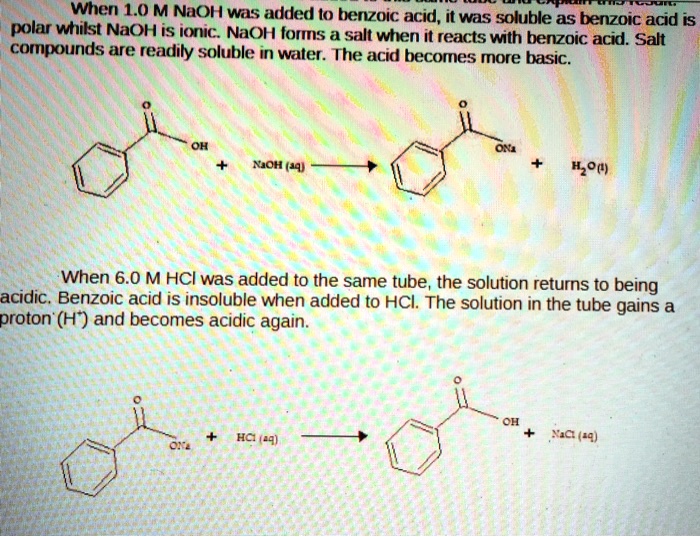
SOLVED: When 1.0 M NaOH was added (0 benzoic acid, it was soluble as benzoic acid is polar wtitst NaOH is ioric; NaOH forms salt when i reacts with berizoic aczo Sad
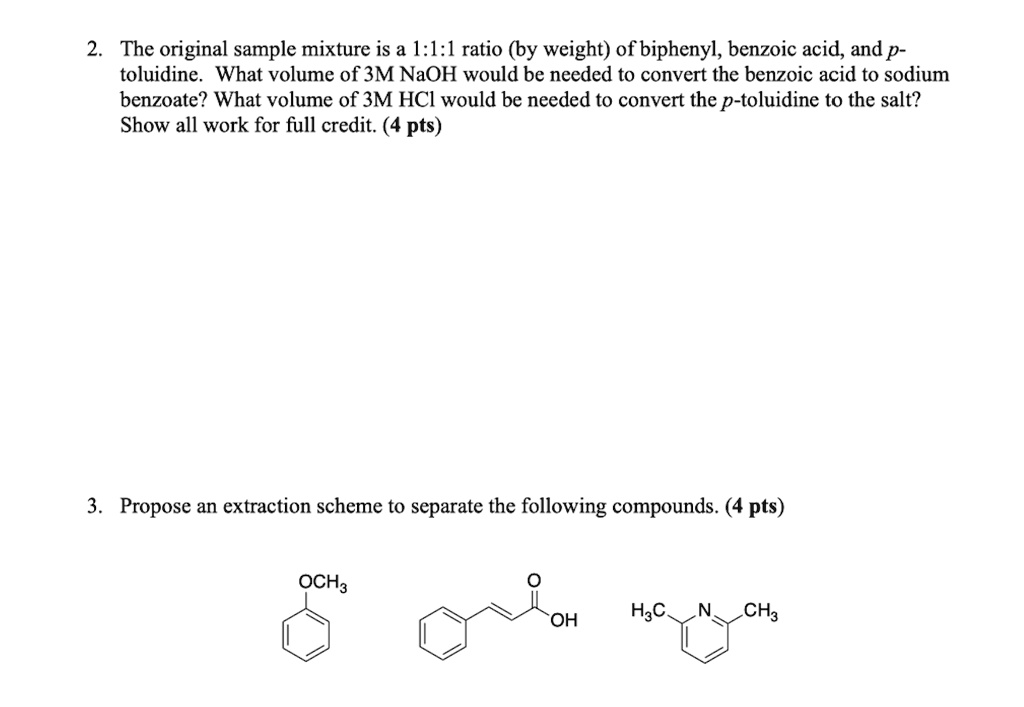
SOLVED: The original sample mixture is a I:l:l ratio (by weight) of biphenyl, benzoic acid, and p- toluidine. What volume of 3M NaOH would be needed to convert the benzoic acid to
✓ Solved: When benzoic acid ( 5 ) is partitioned between diethyl ether and aqueous sodium hydroxide solution...
![When a solution of benzoic acid was titrated with NaOH the pH of the solution when half the acid neutralized was 4.3 . Dissociation constant of the acid is [Use log 2 = 0.3] . When a solution of benzoic acid was titrated with NaOH the pH of the solution when half the acid neutralized was 4.3 . Dissociation constant of the acid is [Use log 2 = 0.3] .](https://dwes9vv9u0550.cloudfront.net/images/3890404/8b3b6f71-c797-41c4-8e11-5cf2fabbd29f.jpg)
When a solution of benzoic acid was titrated with NaOH the pH of the solution when half the acid neutralized was 4.3 . Dissociation constant of the acid is [Use log 2 = 0.3] .

Explain the results for the tube in which 1.0 m naoh was added to benzoic acid. write an equation for - Brainly.com

Experiment 6 Extraction - Separation of Benzoic Acid and Phenanthrene - Experiment 6: Extraction - - Studocu
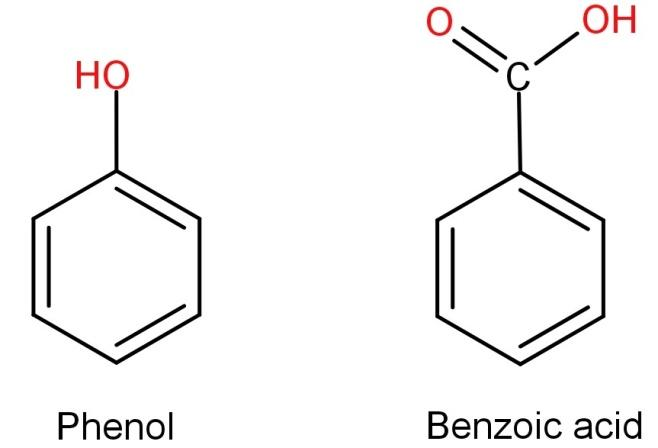
A mixture of phenol and benzoic acid will completely dissolve in an aqueous solution of?(A) HCl(B) NaCl(C) $NaHC{O_3}$ (D) NaOH
✓ Solved: Explain the results for the tube in which 1.0 M NaOH was added to benzoic acid. Write an equation...