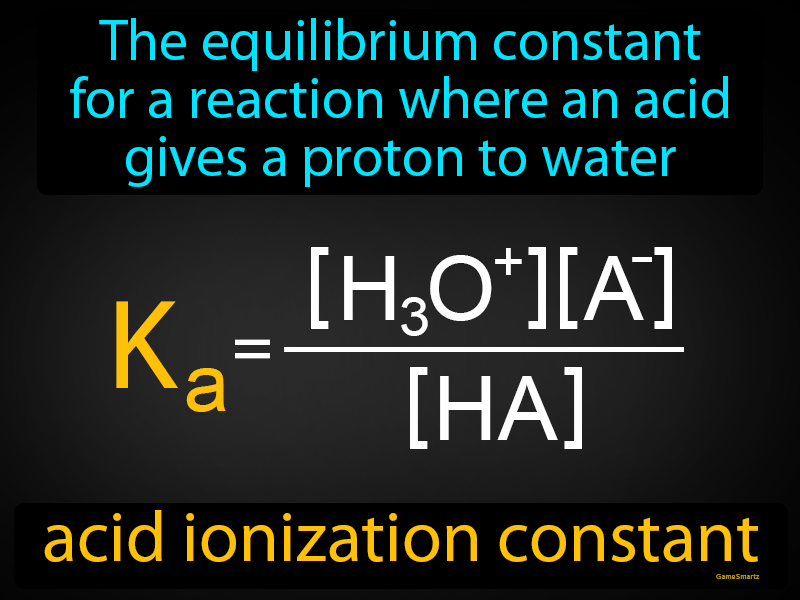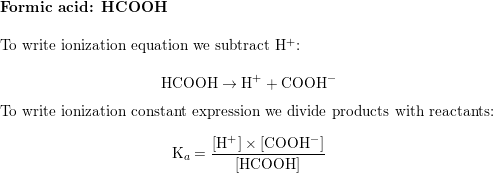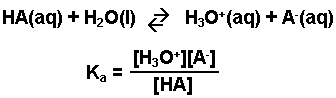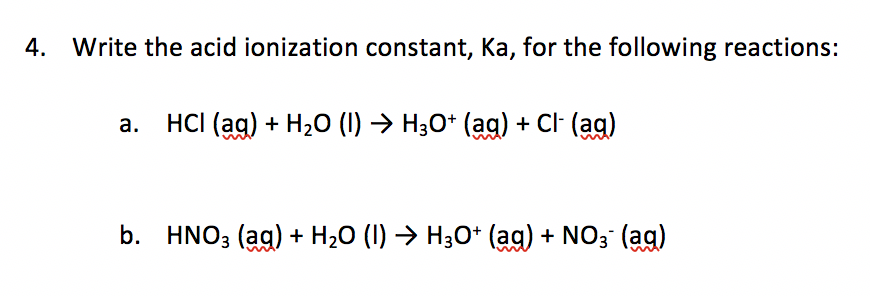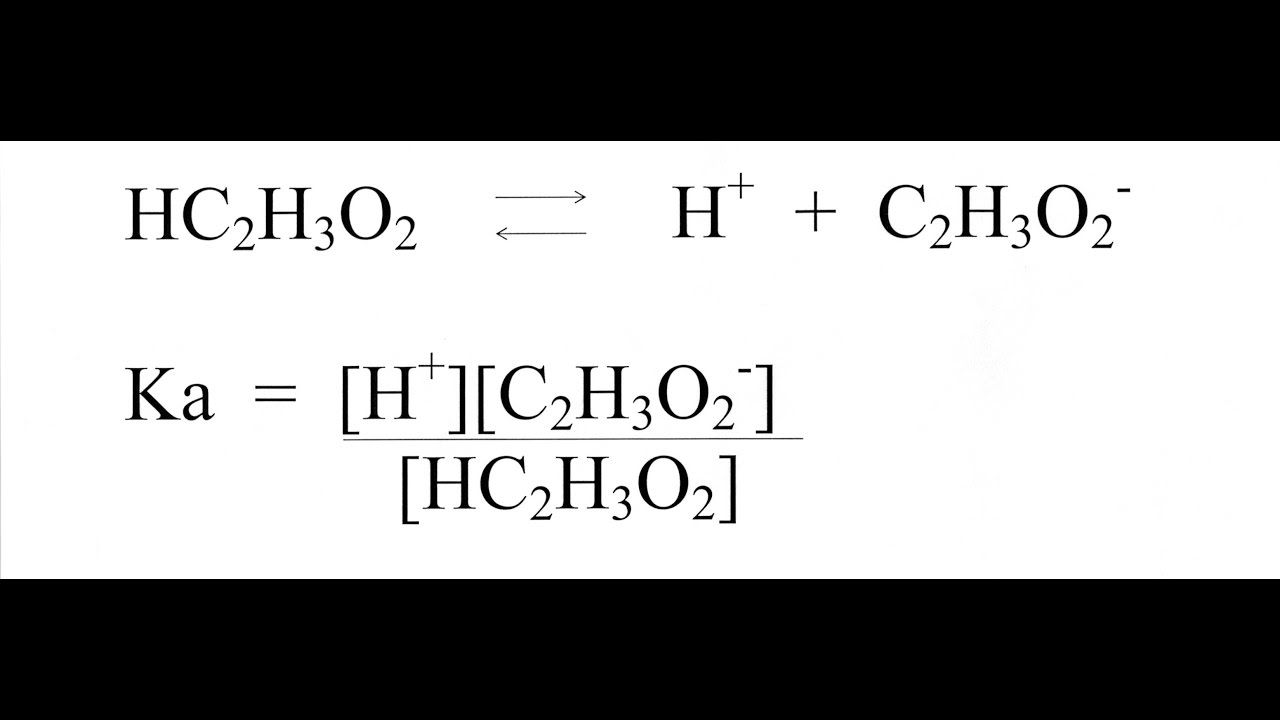What Does the Acid Ionization Constant Tell You? An Organic Chemistry Student Guide | Journal of Chemical Education

Tro: 15.4-15.7) Acid Strength and the Acid Ionization Constant (kA); autoionization of water and pH; find the h3o+ and pH of strong and weak acid solutions; and base solutions Flashcards | Quizlet

8.2) Weak Acids & Bases: Ionization Constants. Percent Ionization for Weak Acids Most weak acids ionize < 50% Percent ionization (p) General Weak Acid: - ppt download

Weak Acids & Acid Ionization Constant Majority of acids are weak. Consider a weak monoprotic acid, HA: The equilibrium constant for the ionization would. - ppt download

The ionization constant of propanoic acid is 1.32 × 10^-5 . Calculate the degree of ionization of the acid in its 0.05 M solution and also its pH . What will be

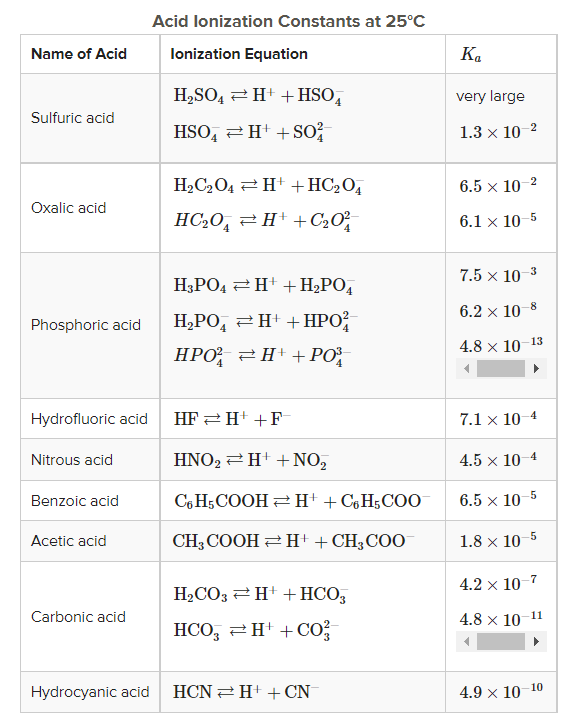
SOLVED: Question 7 1 pts For which of the following reactions is the equilibrium constant called an acid ionization constant; Ka? HCOOHaq) NHz(aq) HCOO-(aq) NH4t(aq) 2 H3Ot(aq) OH-(aq) 2Hz0 HOCIlaq) Hzo Hsot(aq)

Weak Acid Ionization Constants and the Determination of Weak Acid–Weak Base Reaction Equilibrium Constants in the General Chemistry Laboratory | Journal of Chemical Education
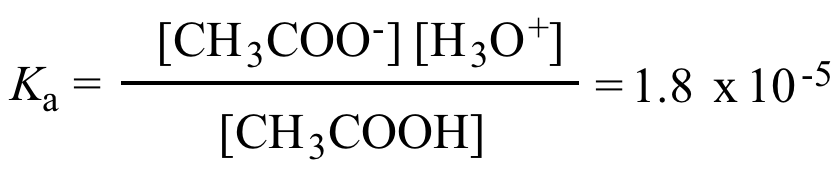
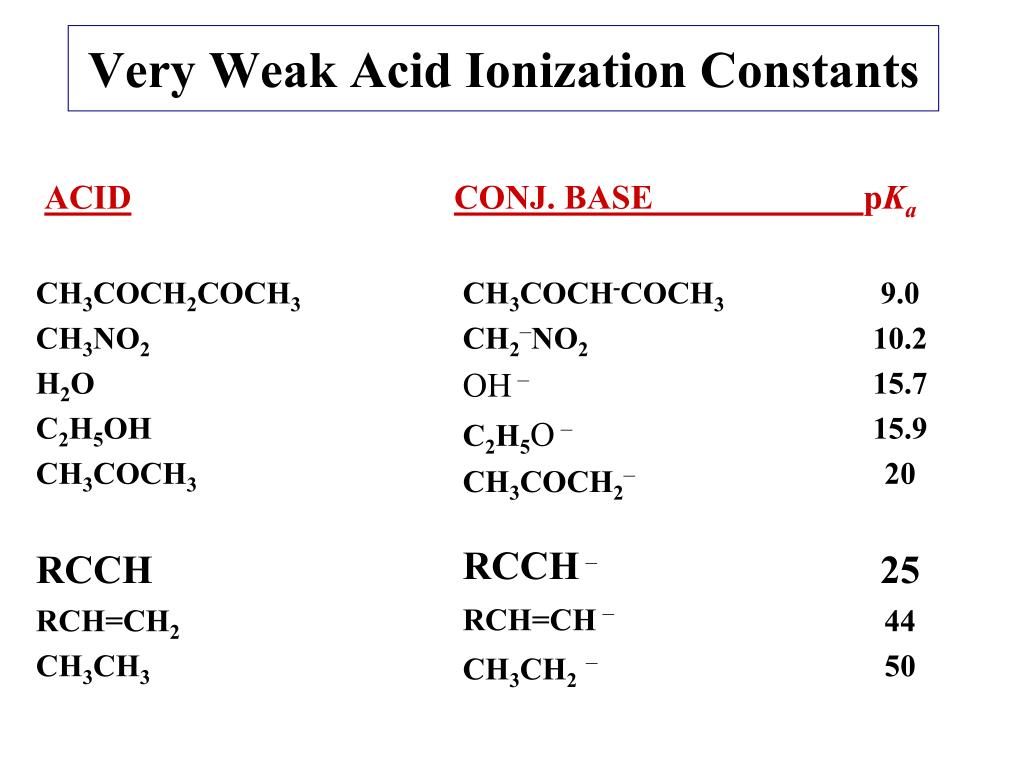
Illustrated Glossary of Organic Chemistry - Acid ionization constant (acid dissociation constant; Ka)

The first and second dissociation constants of an acid H2A are 1.0 × 10^-5 and 5.0 × 10^-10 respectively. The overall dissociation constant of the acid will be: