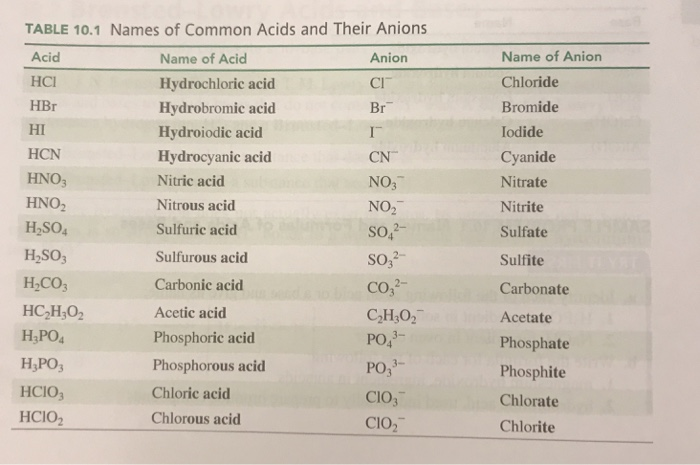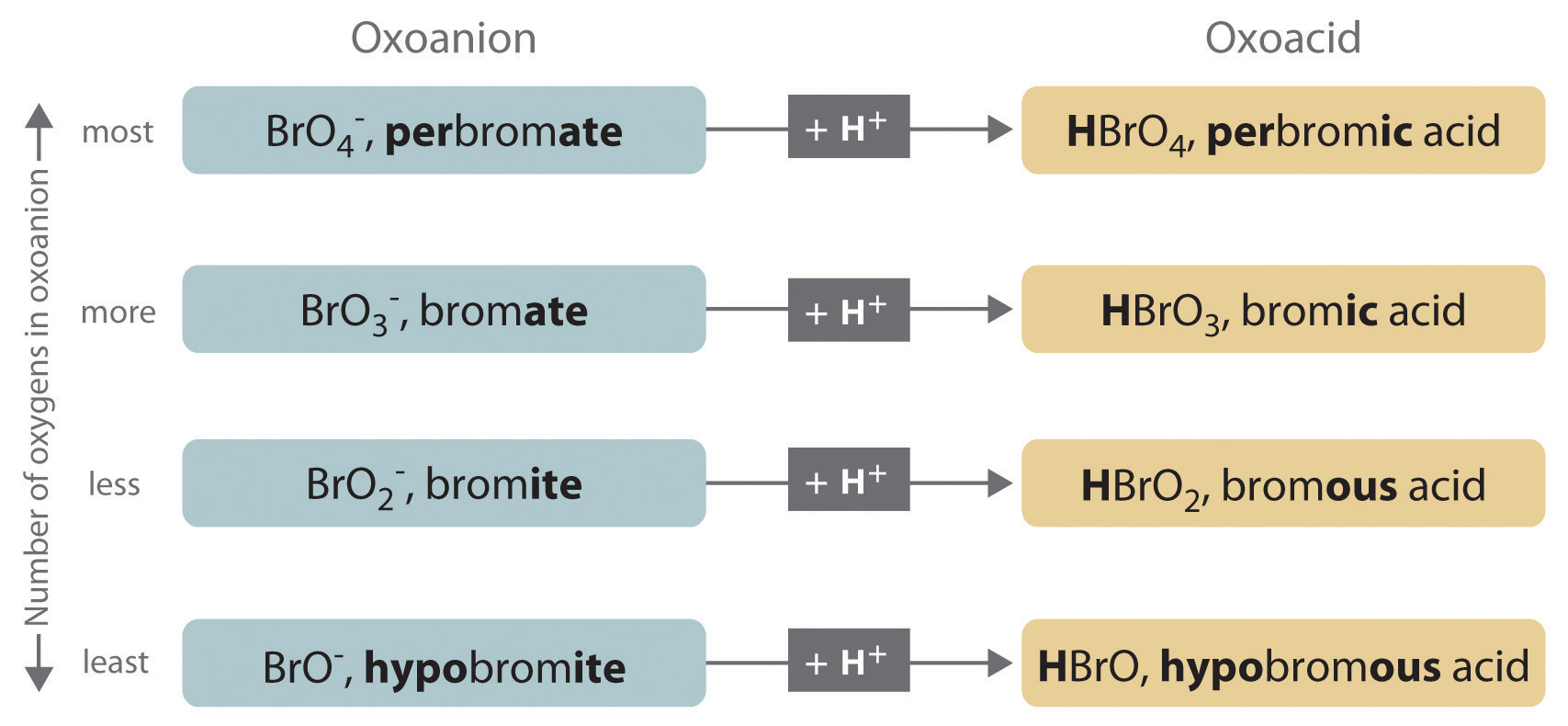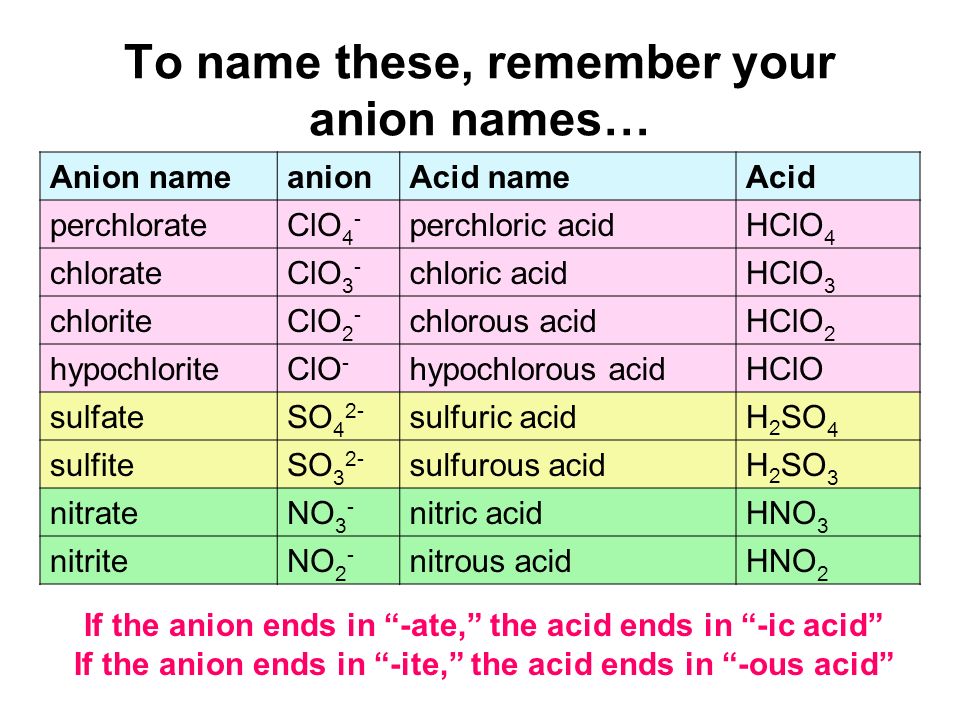
An acid can be defined as a substance that yields hydrogen ions (H + ) when dissolved in water. Simple acid naming: If anion name ends with “-ide” Then. - ppt download

organic chemistry - Why and how carboxylate ion has greater stability than carboxylic acid? - Chemistry Stack Exchange

Loss of a proton from cyanic acid yields the same anion as that obtained by loss of a proton from isocyanic acid. Draw the structure of the anion formed. Loss of a
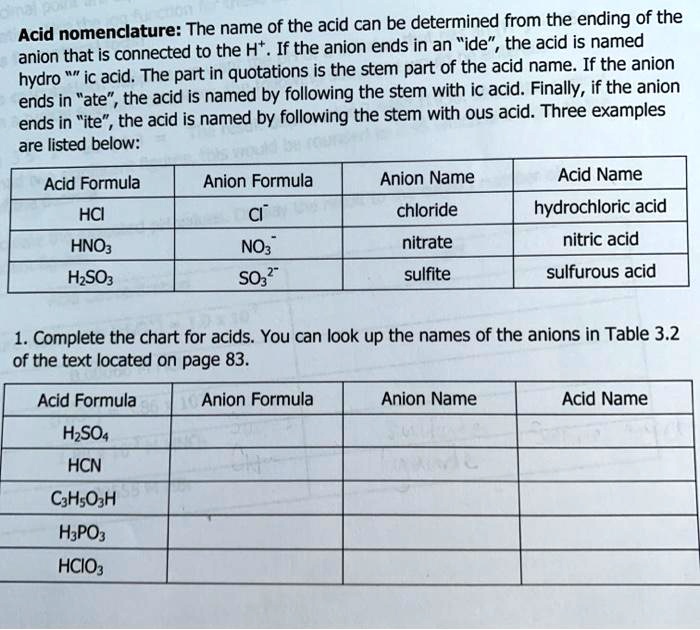
SOLVED: Acid nomenclature: The name of the acid can be determined from the endingof the connected to the H . If the aion ends in an "ide", the acid is named anion